| Management |
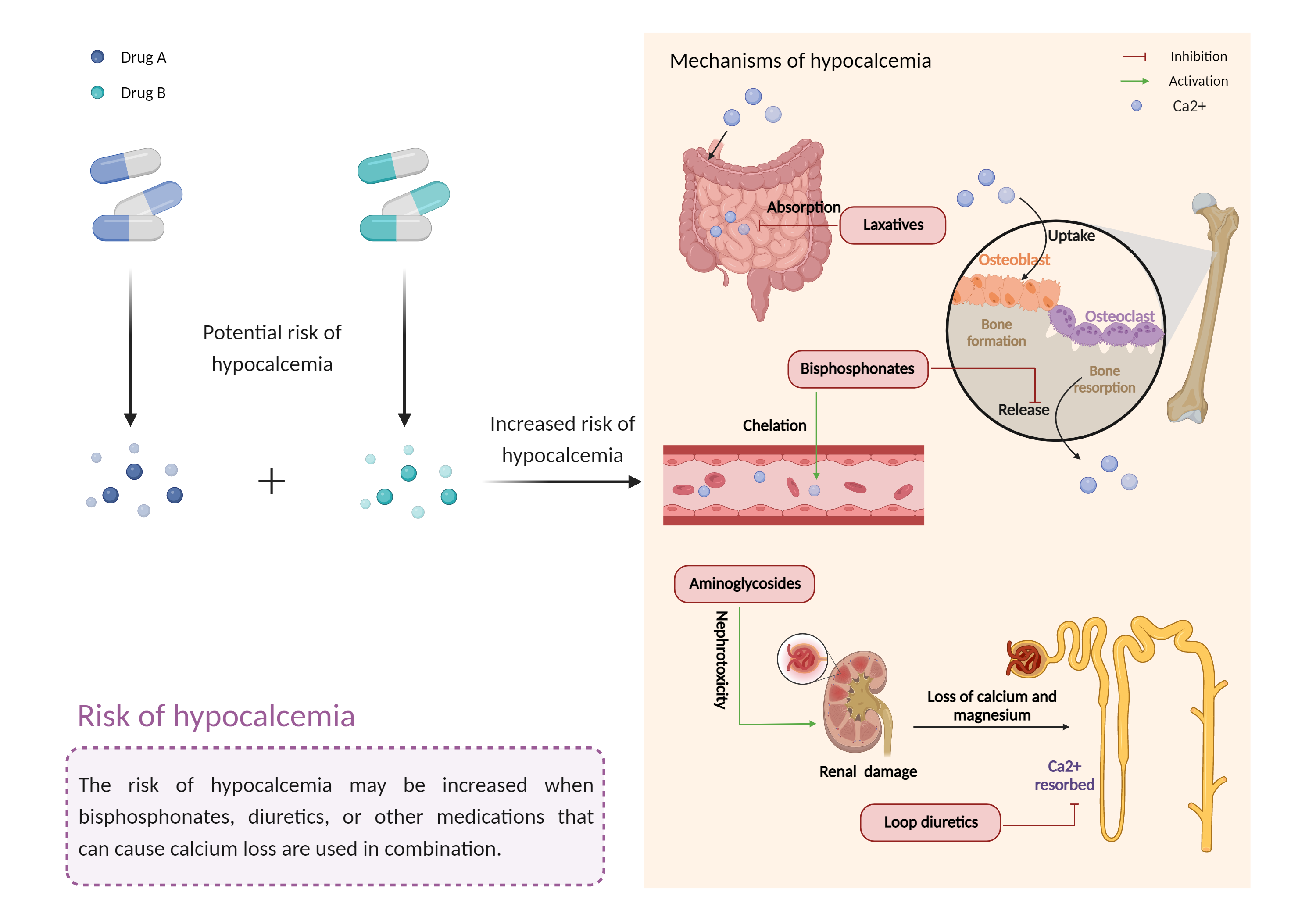
Close monitoring of corrected serum calcium levels is recommended if etelcalcetide is used in combination with other medications known to lower serum calcium, such as denosumab, calcitonin, foscarnet, bisphosphonates, or loop diuretics. It is recommended that the patient's corrected serum calcium level should be measured within one week of initiation or dose adjustment of etelcalcetide and monitored approximately every 4 weeks. In addition, the manufacturer product labeling should be consulted for management guidelines in the event of a reduction in corrected serum calcium levels below the lower limit of the normal range and/or symptoms of hypocalcemia. Patients should also be advised to contact their health care professional if they experience symptoms of hypocalcemia, such as numbness or tingling in the fingers, toes, or around the mouth, muscle spasms, twitches or cramps, seizures, confusion, and irregular heartbeats. |