Details of Drug-Drug Interaction
| Drug General Information (ID: DDIC2K18SO) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Clofibrate | Drug Info | Trabectedin | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antihyperammonia Agents | Alkylating Agents | |||||||
| Structure | |||||||||
| Mechanism of Clofibrate-Trabectedin Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
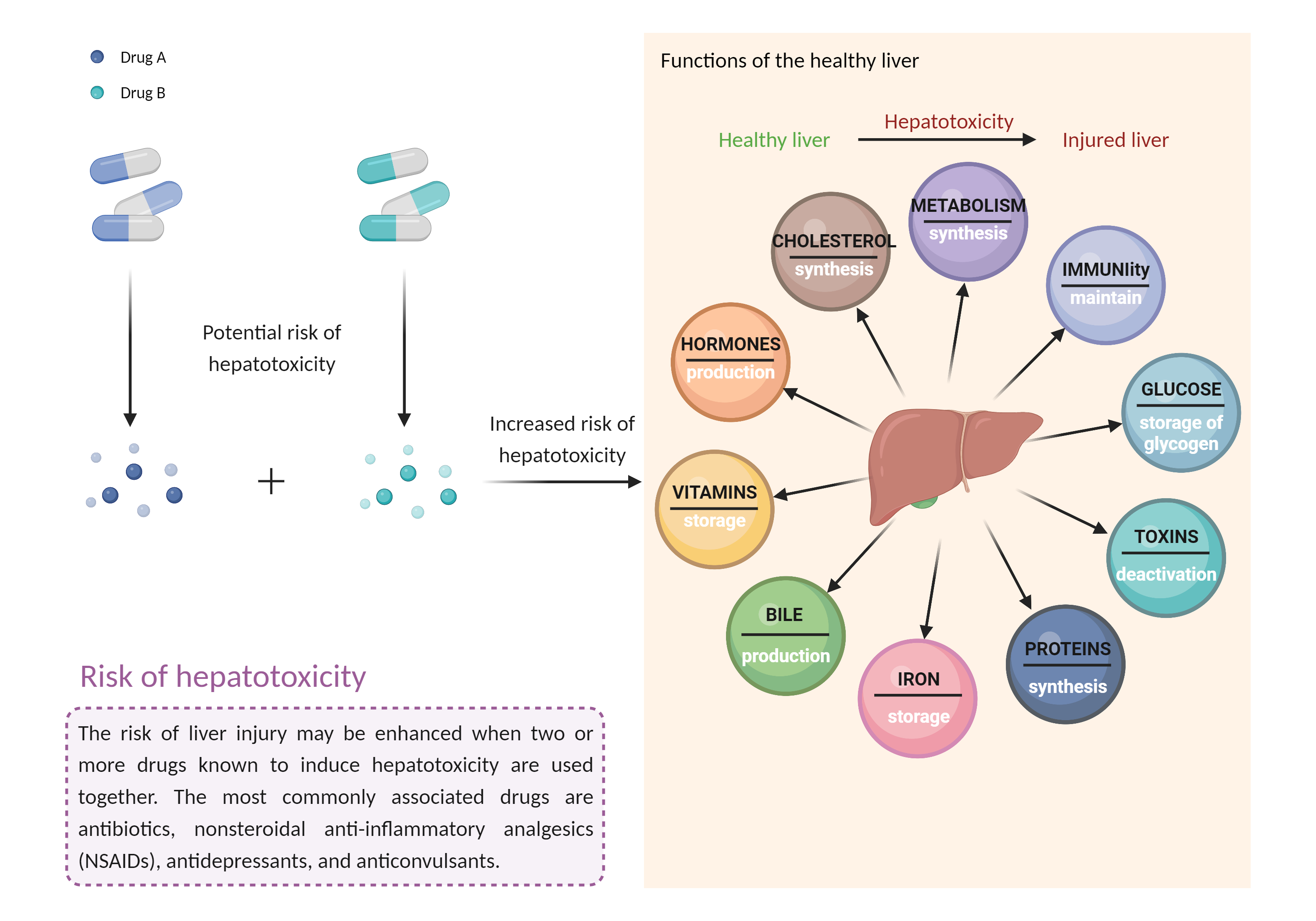
| Increased risk of hepatotoxicity Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Clofibrate | Trabectedin | |||||||
| Mechanism 1 | Hepatotoxicity | Hepatotoxicity | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Hepatotoxicity | ||||||||
| Factor Description | Combination of drugs that can induce hepatotoxicity may increase the risk of liver injury. Symptoms vary depending on the level of exposure and the total extent of liver damage, and may cause few symptoms if the damage is mild, and eventually lead to liver failure in patients with severe damage. | ||||||||
| Mechanism Description |
|
||||||||
| Increased risk of other adverse reactions (Unspecific) Click to Show/Hide Mechanism Graph | |||||||||
| Drug Name | Clofibrate | Trabectedin | |||||||
| Mechanism 2 | Rhabdomyolysis | Rhabdomyolysis | |||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Adverse reactions | ||||||||
| Factor Description | An adverse reaction is an unexpected negative reaction to a medication or treatment that happens even when it's used correctly. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised when trabectedin is used with other agents associated with rhabdomyolysis. Patients should be advised to seek medical attention if they experience unexplained muscle pain, tenderness or weakness, particularly if accompanied by fever, malaise and/or dark colored urine. Caution is advised when trabectedin is used with other agents that are potentially hepatotoxic. Patients should be advised to seek medical attention if they experience potential signs and symptoms of hepatotoxicity such as fever, rash, itching, anorexia, nausea, vomiting, fatigue, malaise, right upper quadrant pain, dark urine, pale stools, and jaundice. Monitoring of alkaline phosphatase, bilirubin, AST, and ALT should occur regularly during trabectedin treatment in accordance with the product labeling, or as often as necessary when clinical symptoms develop. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Cerner Multum, Inc. "UK Summary of Product Characteristics.". | ||||||||||||||||||
| 2 | Product Information. Yondelis (trabectedin). Janssen Pharmaceuticals, Titusville, NJ. | ||||||||||||||||||
