Details of Drug-Drug Interaction
| Drug General Information (ID: DDIBX68F1U) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Siponimod | Drug Info | Human adenovirus e serotype 4 strain cl-68578 antigen | Drug Info | |||||
| Drug Type | Small molecule | Vaccine | |||||||
| Therapeutic Class | Selective Immunosuppressants | Vaccine | |||||||
| Mechanism of Siponimod-Human adenovirus e serotype 4 strain cl-68578 antigen Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
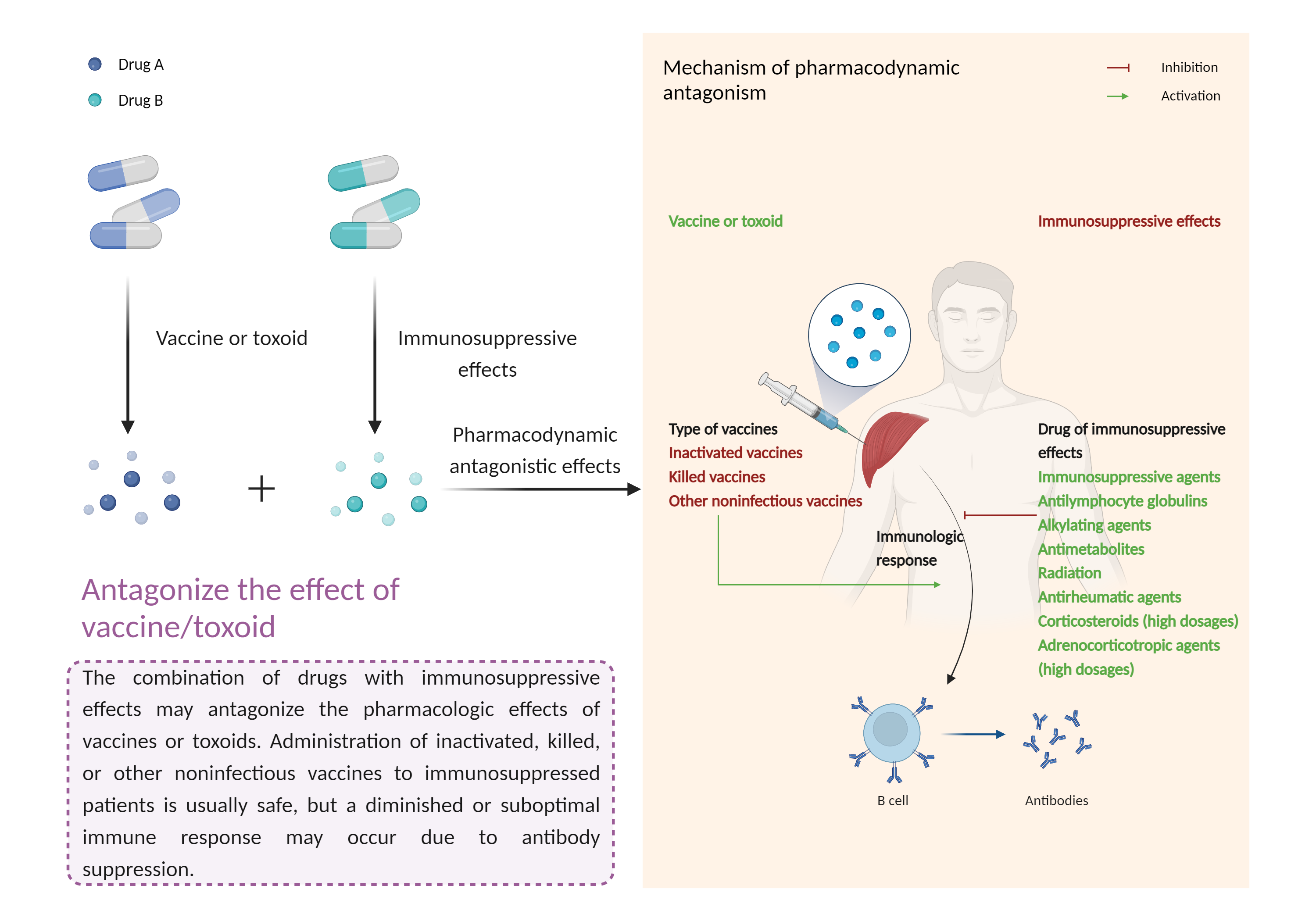
| Antagonize the effect of vaccine/toxoid Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Siponimod | Human adenovirus e serotype 4 strain cl-68578 antigen | |||||||
| Mechanism | Immunosuppressive effects | Vaccine or toxoid | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Vaccine/toxoid | ||||||||
| Factor Description | The beneficial effects of the vaccine/toxinoid may be reduced, leading to a diminished or suboptimal immune response. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | The use of live attenuated vaccines should be avoided during and for up to 4 weeks after siponimod treatment, and siponimod should be paused 1 week prior to and until 4 weeks after a planned vaccination. Patients without a healthcare professional confirmed history of varicella or without documentation of a full course of vaccination against varicella-zoster virus (VZV) should be tested for antibodies to VZV before initiating treatment with siponimod. For antibody-negative patients, a full course of vaccination with varicella vaccine is recommended prior to treatment with siponimod. In patients who have recently been vaccinated, siponimod therapy should be postponed for 4 weeks to allow the full effect of vaccination to occur. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Cerner Multum, Inc. "Australian Product Information.". | ||||||||||||||||||
| 2 | Product Information. Mayzent (siponimod). Novartis Pharmaceuticals, East Hanover, NJ. | ||||||||||||||||||
