Details of Drug-Drug Interaction
| Drug General Information (ID: DDIB3HUMI1) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Octreotide | Drug Info | Lutetium Lu 177 dotatate | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Somatostatin/Somatostatin Analogs | Therapeutic Radiopharmaceuticals | |||||||
| Structure | |||||||||
| Mechanism of Octreotide-Lutetium Lu 177 dotatate Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
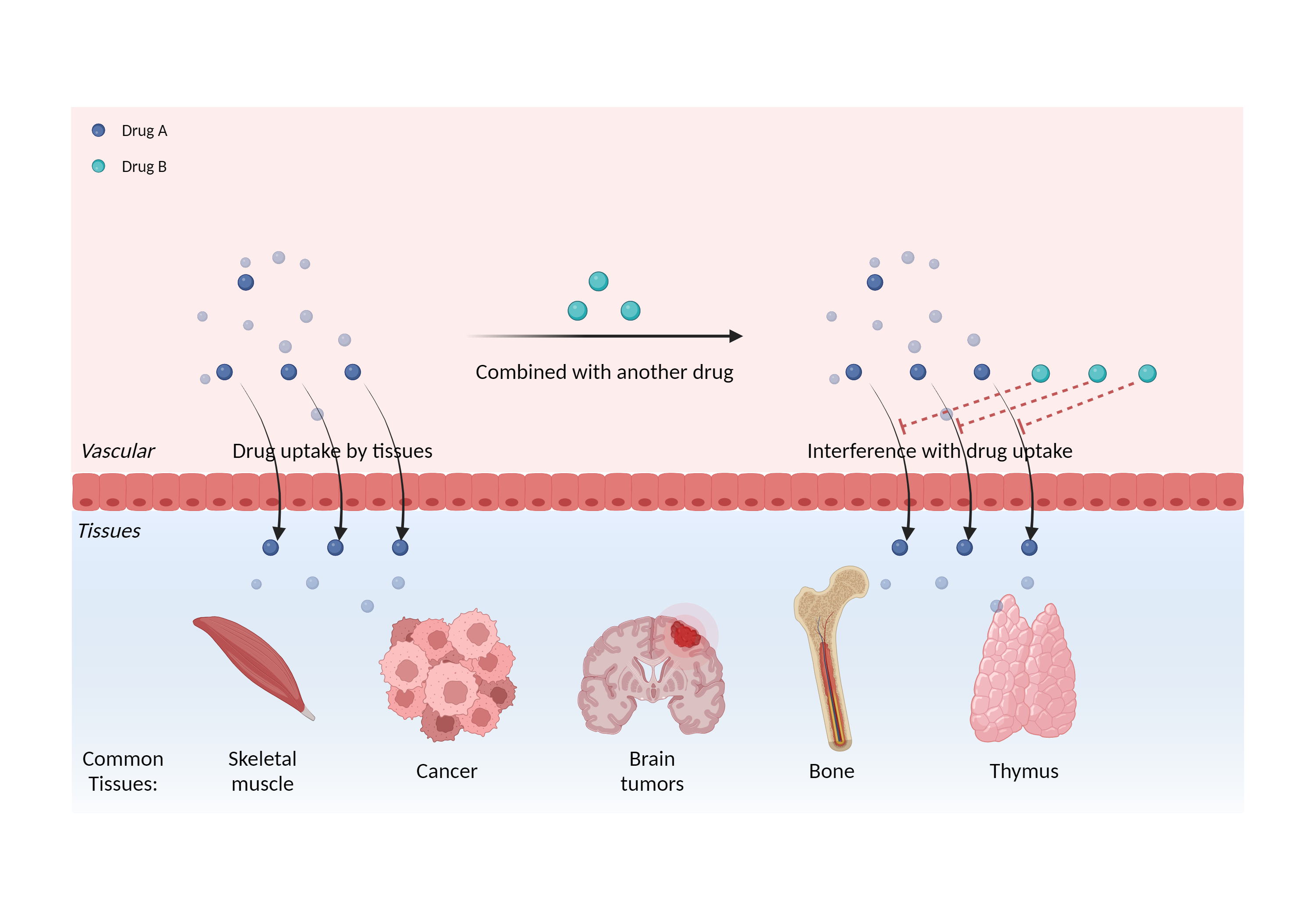
| Interference of cell/tissue uptake Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Octreotide | Lutetium Lu 177 dotatate | |||||||
| Mechanism | Somatostatin analog | Affects somatostatin analog diagnostic test results | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Pharmacokinetics | ||||||||
| Factor Description | Alters the distribution of the drug, leading to changes in the plasma concentration of the drug and affecting the pharmacological effects. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Long-acting formulations of somatostatin analogs should be discontinued for at least 4 weeks and short-acting formulations for at least 24 hours prior to each dose of lutetium Lu 177 dotatate. During treatment with lutetium Lu 177 dotatate, long-acting octreotide should be administered intramuscularly between 4 to 24 hours after each dose of lutetium Lu 177 dotatate (but not within 4 weeks of each subsequent dose of lutetium Lu 177 dotatate), while short-acting octreotide may be given as needed for symptomatic management (but not within 24 hours before each subsequent dose of lutetium Lu 177 dotatate). Following completion of lutetium Lu 177 dotatate treatment, long-acting octreotide should be continued every 4 weeks until disease progression or up to 18 months following treatment initiation. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Lutathera (lutetium Lu 177 dotatate). Advanced Accelerator Applications, New York, NY. | ||||||||||||||||||
