Details of Drug-Drug Interaction
| Drug General Information (ID: DDIATKZ5WY) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Ritonavir | Drug Info | Tipranavir | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Anti-Hiv Agents | Anti-Hiv Agents | |||||||
| Structure | |||||||||
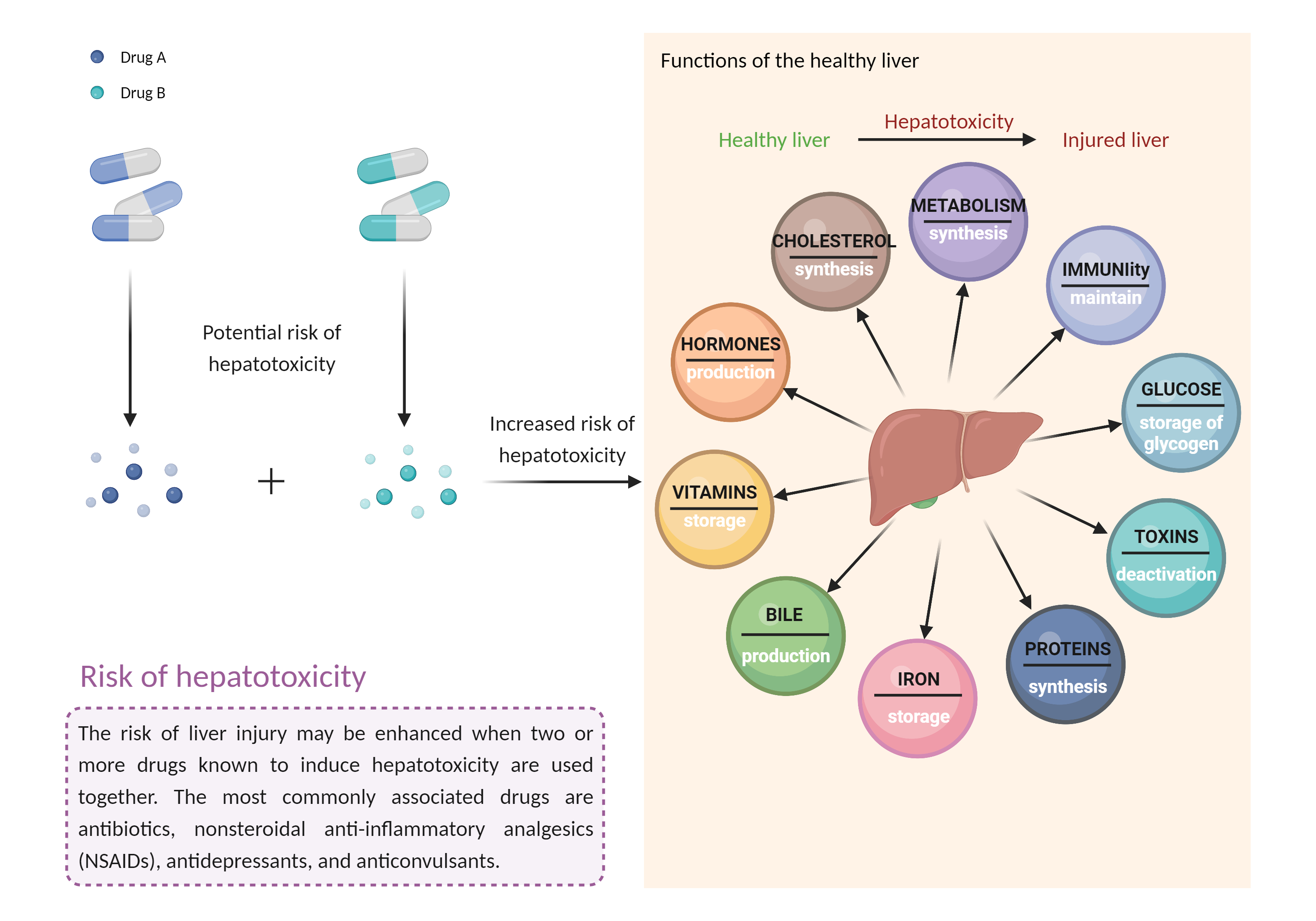
| Mechanism of Ritonavir-Tipranavir Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Increased risk of hepatotoxicity Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Ritonavir | Tipranavir | |||||||
| Mechanism | Hepatotoxicity | Hepatotoxicity | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Hepatotoxicity | ||||||||
| Factor Description | Combination of drugs that can induce hepatotoxicity may increase the risk of liver injury. Symptoms vary depending on the level of exposure and the total extent of liver damage, and may cause few symptoms if the damage is mild, and eventually lead to liver failure in patients with severe damage. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Close monitoring of hepatic function is recommended in patients treated with tipranavir and ritonavir. Particular caution is warranted in patients with chronic hepatitis B or C coinfection, as they may have an increased risk of hepatotoxicity. Patients should be advised to notify their physician if they experience signs and symptoms of hepatotoxicity such as fever, rash, anorexia, nausea, vomiting, fatigue, right upper quadrant pain, dark urine, and jaundice. | ||||||||
