Details of Drug-Drug Interaction
| Drug General Information (ID: DDIAILQKY1) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Dapsone | Drug Info | Zalcitabine | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antiinflammatory Agents | Anti-Hiv Agents | |||||||
| Structure | |||||||||
| Mechanism of Dapsone-Zalcitabine Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
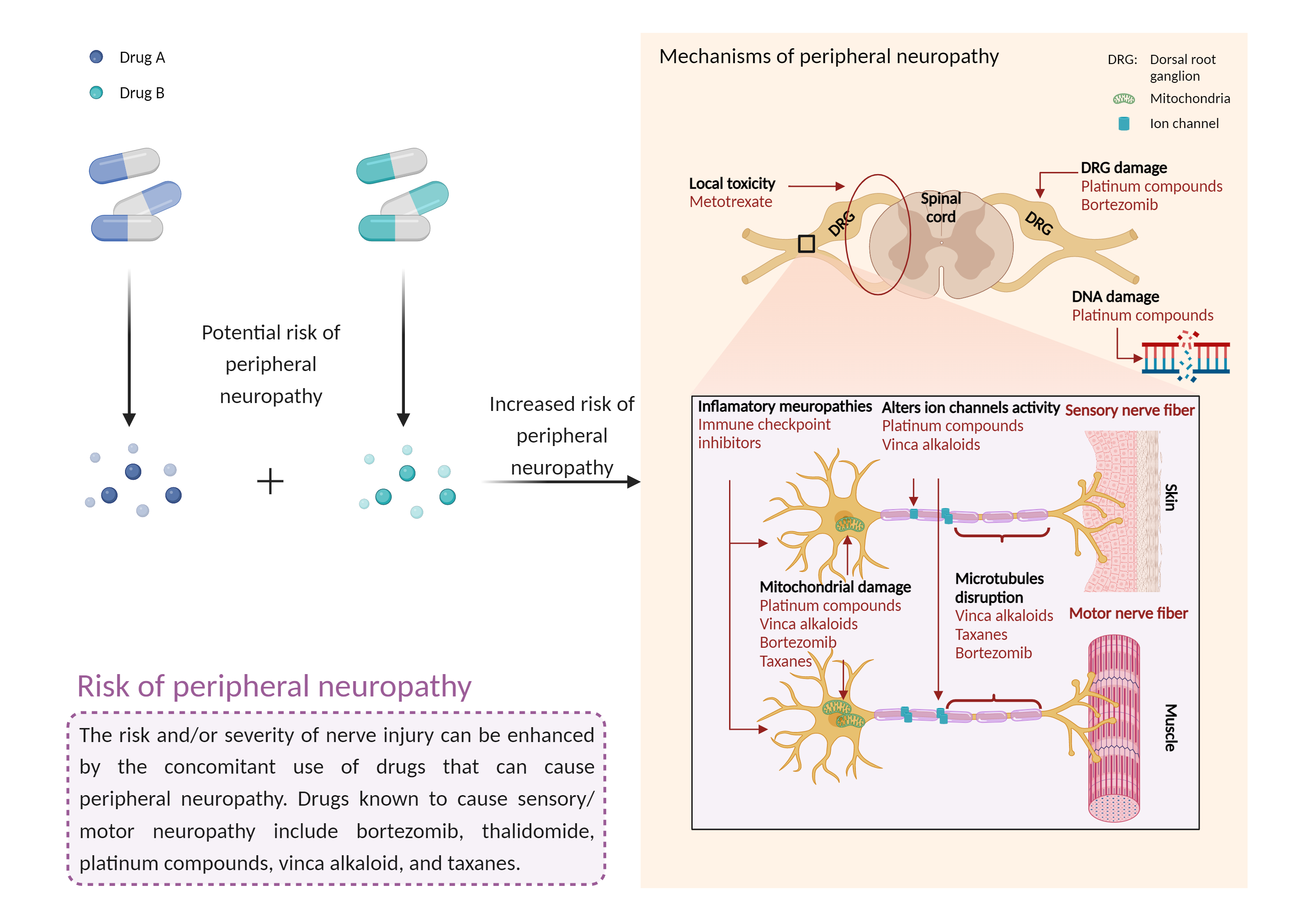
| Increased risk of peripheral neuropathy Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Dapsone | Zalcitabine | |||||||
| Mechanism | Peripheral neuropathy | Peripheral neuropathy | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Peripheral neuropathy | ||||||||
| Factor Description | Peripheral neuropathy is a disorder of the peripheral nerves that can cause sensory and motor symptoms, and even paralysis in severe cases. Signs and symptoms of peripheral neuropathy may include: gradual numbness, tingling or prickling in the feet or hands that can spread up the legs and arms; sharp, stinging, pricking or burning sensations; extreme sensitivity to touch; pain during activities that should not cause pain; lack of coordination and falls; muscle weakness; and paralysis if the motor nerves are affected. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Use of zalcitabine with other drugs that have the potential to cause peripheral neuropathy should be avoided whenever possible. Otherwise, careful monitoring is recommended for symptoms of neuropathy such as burning, tingling, pain, numbness, or weakness in the extremities, particularly in patients with a low CD4 cell count or diabetes. Since the development of peripheral neuropathy appears to be dose-related, the recommended dosage of zalcitabine should not be exceeded. Patients should be advised to promptly discontinue zalcitabine therapy and contact their physician if neuropathy develops. Therapy may be reinstituted following resolution of neuropathy symptoms, but dosage should be reduced to one-half the initially recommended dosage. Zalcitabine should be permanently discontinued in patients who develop severe peripheral neuropathy during treatment. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Argov Z, Mastaglia FL "Drug-induced peripheral neuropathies." Br Med J 1 (1979): 663-6. [PMID: 219931] | ||||||||||||||||||
| 2 | Product Information. Hivid (zalcitabine). Roche Laboratories, Nutley, NJ. | ||||||||||||||||||
