| Mechanism of Inotuzumab ozogamicin-Denosumab Interaction
(Severity Level: Moderate)
|
|
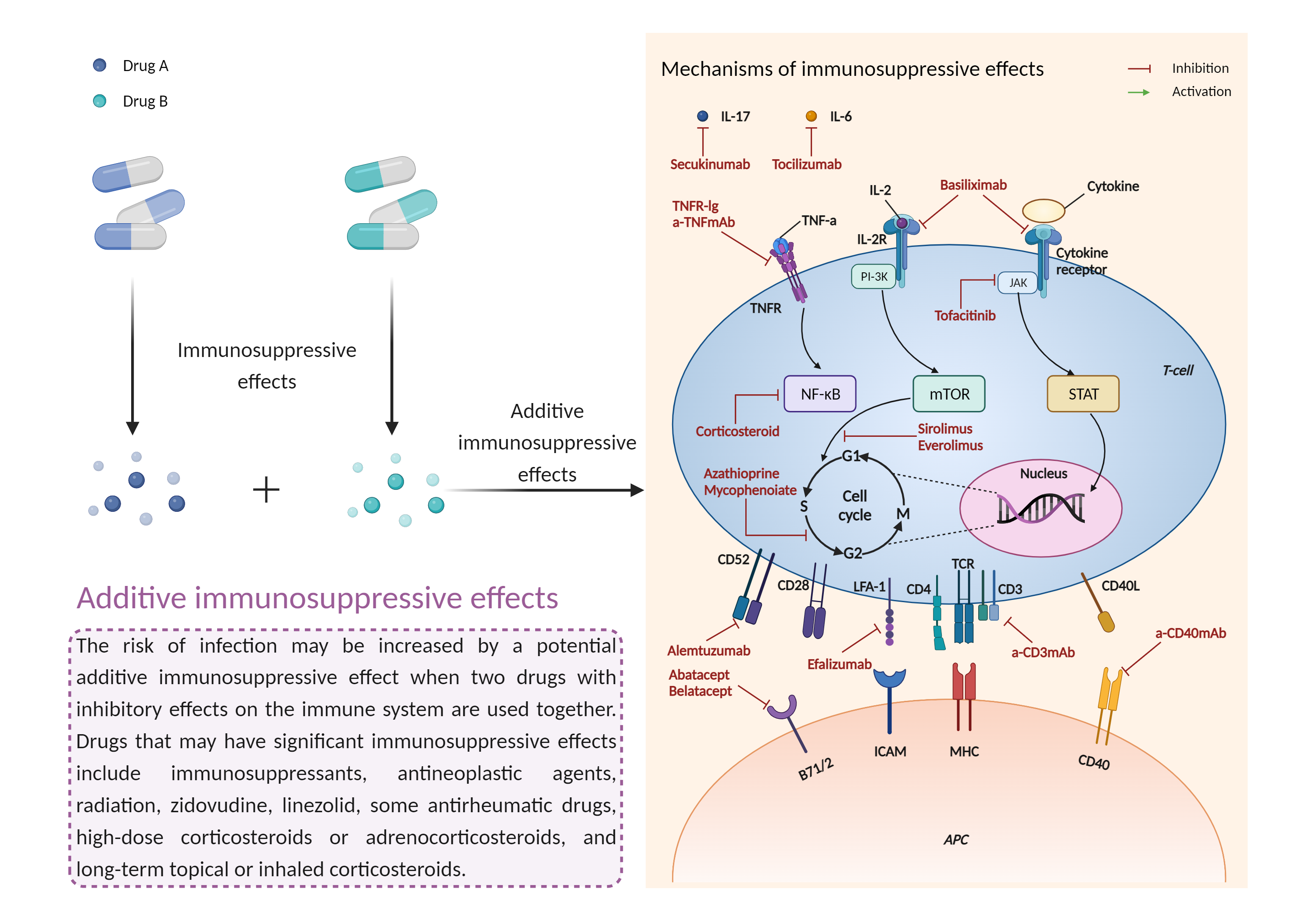
Additive immunosuppressive effects
Click to Show/Hide Mechanism Graph
|
 |
| Drug Name |
Inotuzumab ozogamicin |
Denosumab |
|
Mechanism 1
|
Immunosuppressive effects |
Immunosuppressive effects |
| Key Mechanism Factor 1 |
| Factor Name |
Immunosuppressive effects |
| Factor Description |
Immunosuppression is when your immune system is not functioning as it should. The immune system is made up of cells, tissues and organs that help the body fight off infections. If the immune system is suppressed, an infection that your body was able to control may become serious or even fatal. |
| Mechanism Description |
- Additive immunosuppressive effects by the combination of Inotuzumab ozogamicin and Denosumab
|
|
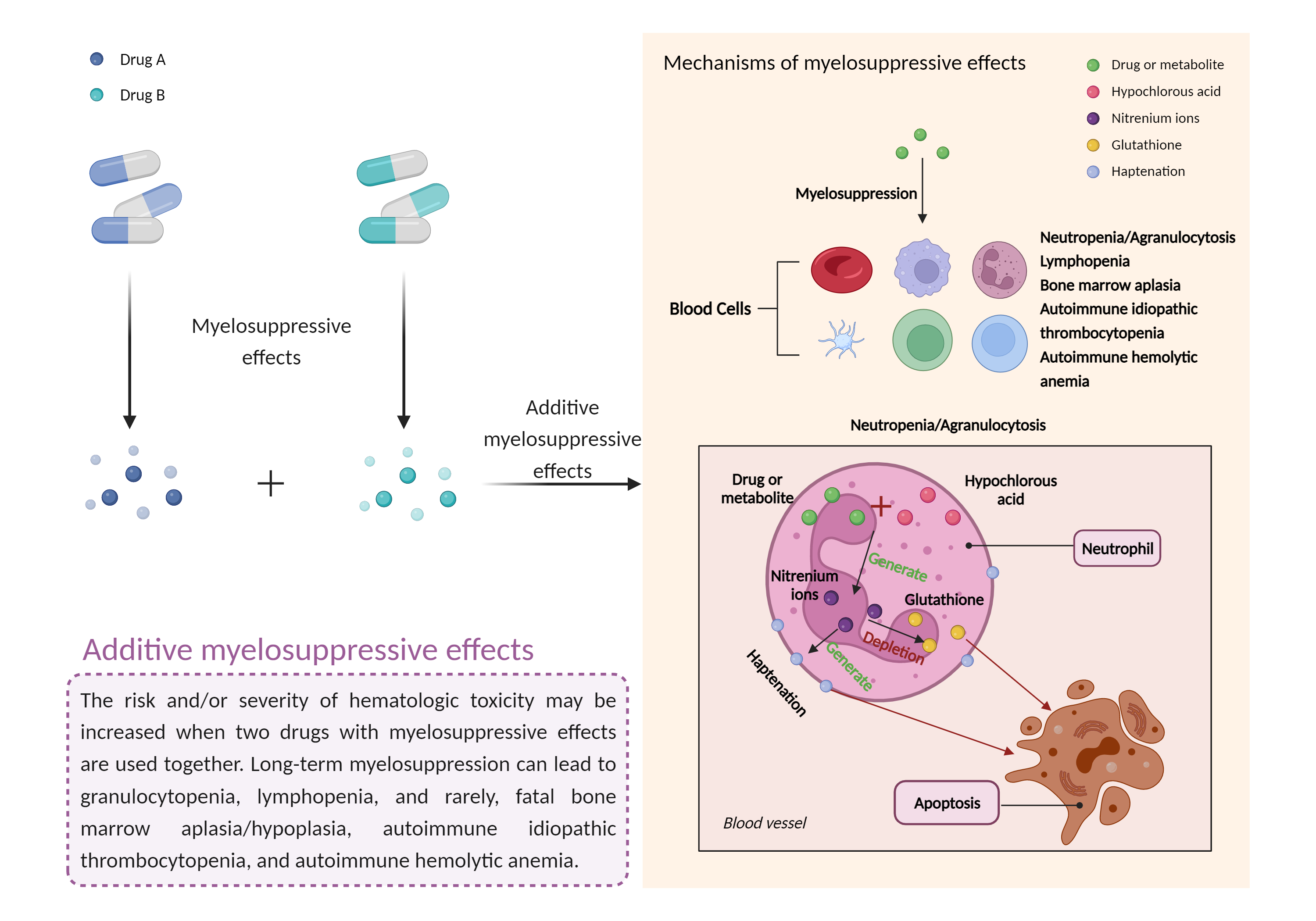
Additive myelosuppressive effects
Click to Show/Hide Mechanism Graph
|
 |
| Drug Name |
Inotuzumab ozogamicin |
Denosumab |
|
Mechanism 2
|
Myelosuppressive effects |
Myelosuppressive effects |
| Key Mechanism Factor 2 |
| Factor Name |
Myelosuppressive effects |
| Factor Description |
Myelosuppression, also known as bone marrow suppression, is a decrease in bone marrow activity that leads to a decrease in the production of blood cells. Some blood cell disorders include: erythrocytopenia (anemia), leukopenia (neutropenia), and thrombocytopenia (thrombocytopenia). |
| Mechanism Description |
- Additive myelosuppressive effects by the combination of Inotuzumab ozogamicin and Denosumab
|


