Details of Drug-Drug Interaction
| Drug General Information (ID: DDI9MGBELO) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Triethylenetetramine | Drug Info | Iron protein succinylate | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Chelating Agents | Iron Supplement | |||||||
| Mechanism of Triethylenetetramine-Iron protein succinylate Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
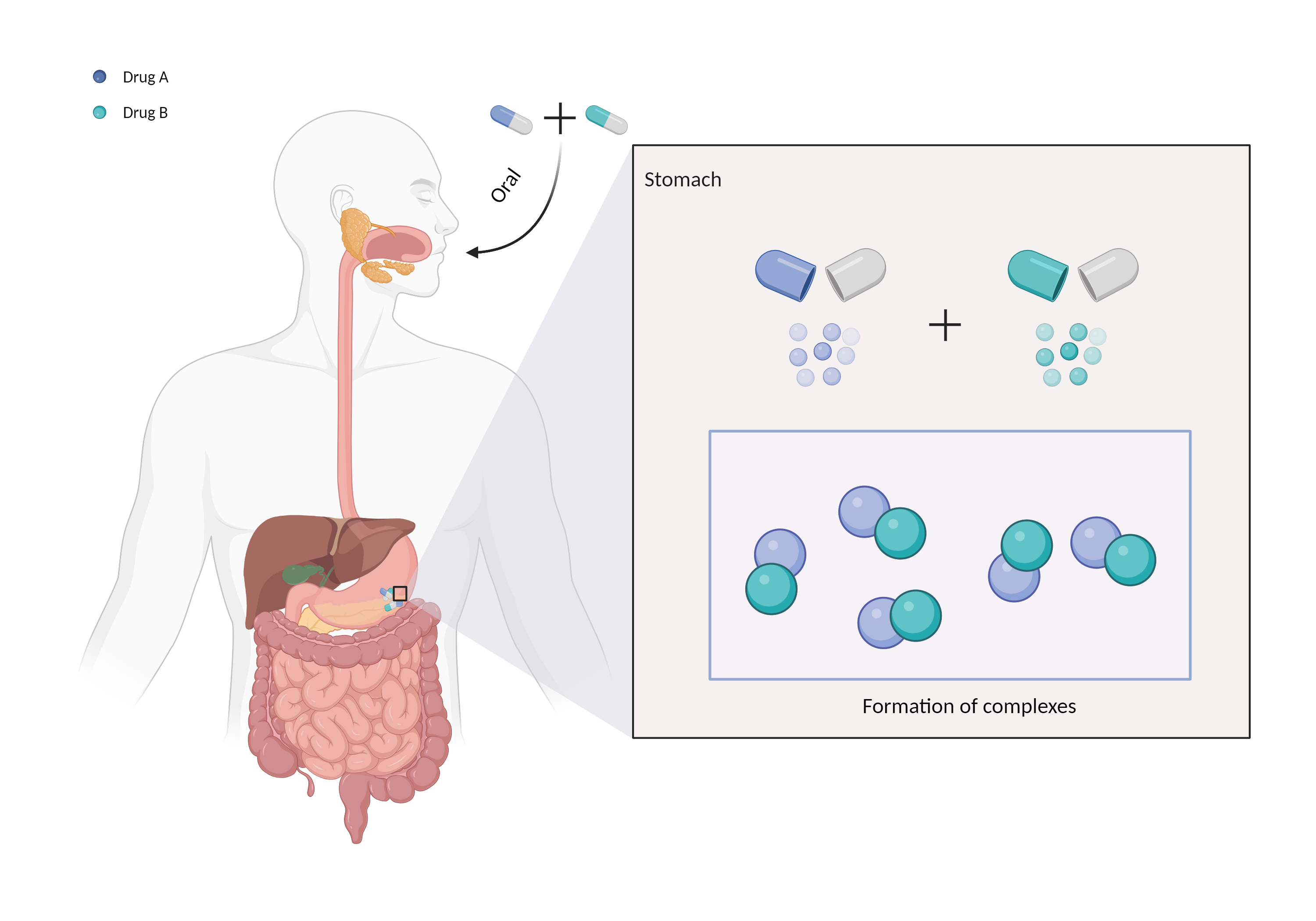
| Complex formation Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Triethylenetetramine | Iron protein succinylate | |||||||
| Mechanism | Binds to polyvalent cations | Polyvalent cations | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Chelation | ||||||||
| Factor Description | Chelation is a direct drug interaction that usually involves the formation of dimers or trimers, resulting in larger complexes and poorer absorption. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | In general, mineral supplements and other products containing minerals should not be used in patients treated with trientine. However, iron deficiency may develop, particularly in children and menstruating or pregnant women. If necessary, iron may be given in short courses, provided that administration times are separated by at least two hours. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Syprine (trientine). Aton Pharma, Lawrenceville, NJ. | ||||||||||||||||||
