Details of Drug-Drug Interaction
| Drug General Information (ID: DDI9M2O7K8) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Anagrelide | Drug Info | Cilostazol | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antithrombotic Agents | Vasodilator Agents | |||||||
| Structure | |||||||||
| Mechanism of Anagrelide-Cilostazol Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
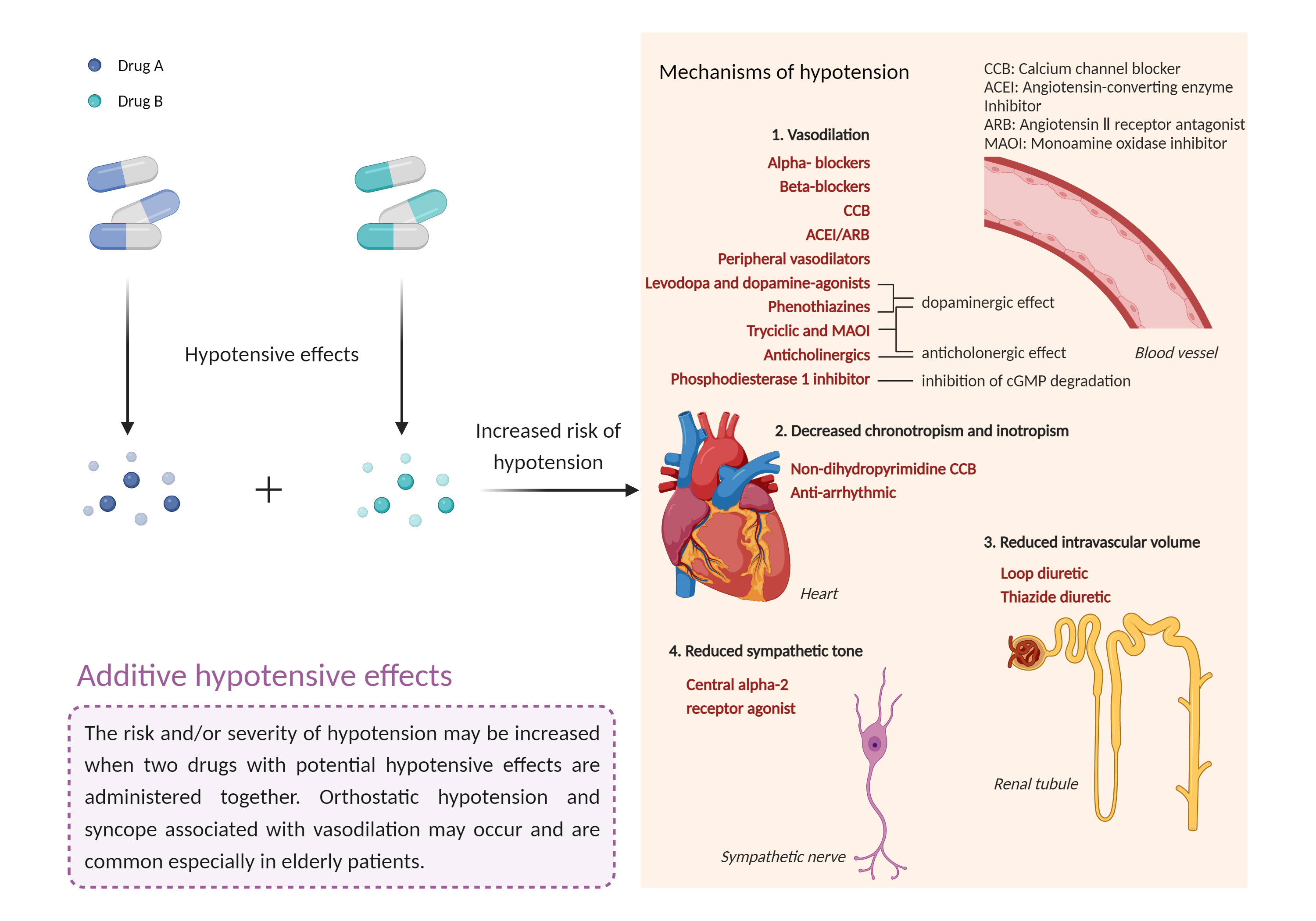
| Additive hypotensive effects Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Anagrelide | Cilostazol | |||||||
| Mechanism 1 |
Hypotensive effects Phosphodiesterase 3 Inhibitor |
Hypotensive effects Phosphodiesterase 3 Inhibitor |
|||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Phosphodiesterase 3 | Structure Sequence | |||||||
| Protein Family | Cyclic nucleotide phosphodiesterase family | ||||||||
| Protein Function |
Cyclic nucleotide phosphodiesterase with specificity for the second messengers cAMP and cGMP, which are key regulators of many important physiological processes (PubMed:1315035, PubMed:8695850, PubMed:8155697, PubMed:25961942). Has also activity toward cUMP (PubMed:27975297). Independently of its catalytic activity it is part of an E2/17beta-estradiol-induced pro-apoptotic signaling pathway. E2 stabilizes the PDE3A/SLFN12 complex in the cytosol, promoting the dephosphorylation of SLFN12 and activating its pro-apoptotic ribosomal RNA/rRNA ribonuclease activity. This apoptotic pathway might be relevant in tissues with high concentration of E2 and be for instance involved in placenta remodeling (PubMed:31420216, PubMed:34707099).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
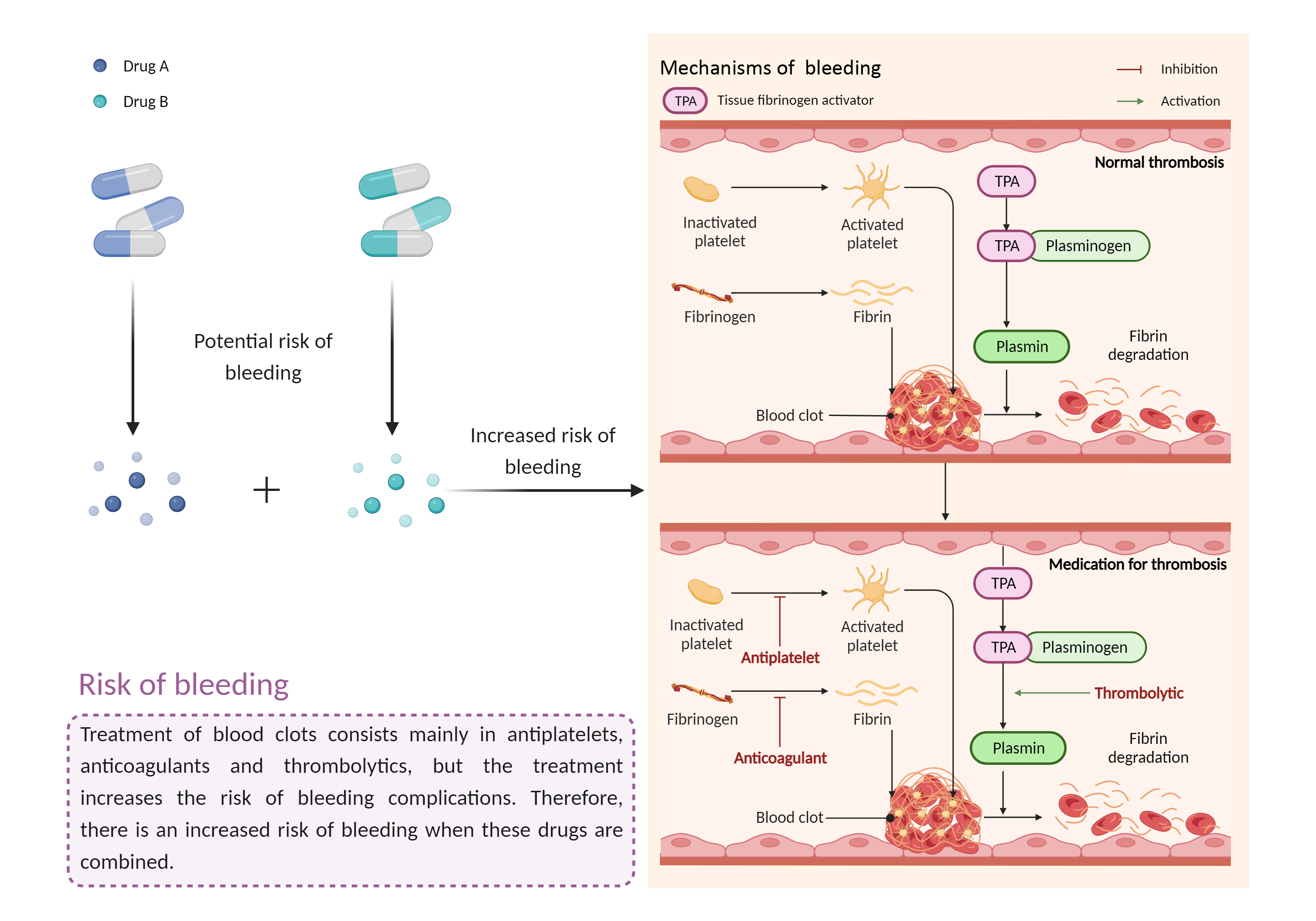
| Increased risk of bleeding Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Anagrelide | Cilostazol | |||||||
| Mechanism 2 |
Risk of bleeding Antiplatelet |
Risk of bleeding Antiplatelet |
|||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Bleeding | ||||||||
| Factor Description | Patients may be at risk for bleeding when the outcome of a disease or disease treatment confounds the standard mechanisms for maintaining hemostasis. Signs or symptoms of abnormal bleeding include: bleeding that takes a long time to stop (including nosebleeds, bleeding gums, bleeding from cuts and abrasions, and menstrual bleeding); severe unexplained bruising, or bruising that becomes larger; blood in the urine or stool, etc. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised if anagrelide is coadministered with other inotropes or inhibitors. The risk of potentially additive cardiovascular adverse effects should be considered. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Agrylin (anagrelide). Roberts Pharmaceutical Corporation, Eatontown, NJ. | ||||||||||||||||||
