Details of Drug-Drug Interaction
| Drug General Information (ID: DDI8HIM4ZG) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Tramadol | Drug Info | Ondansetron | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Analgesics | Antiemetics | |||||||
| Structure | |||||||||
| Mechanism of Tramadol-Ondansetron Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Additive serotonergic effects Click to Show/Hide Mechanism Graph | |||||||||
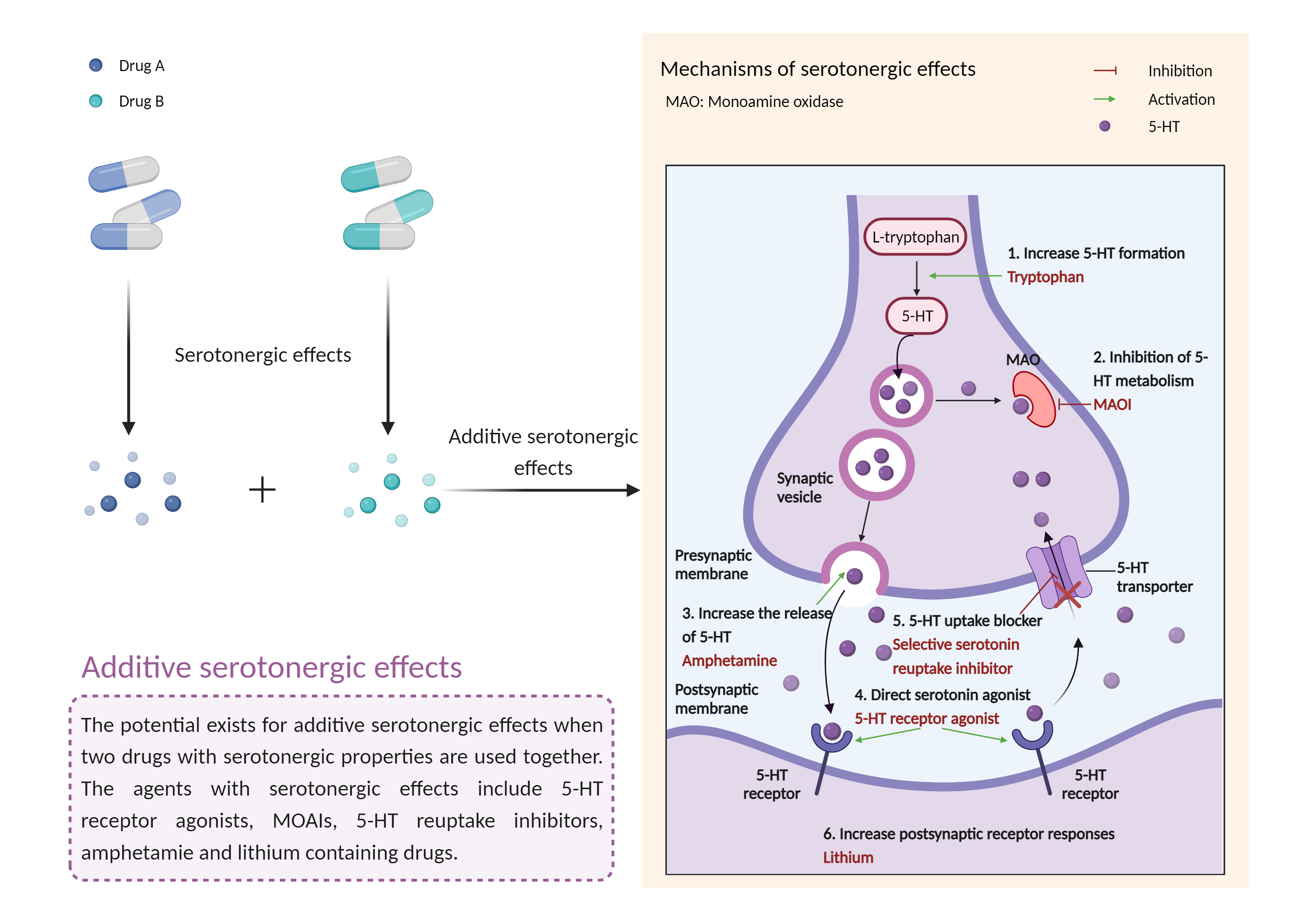 |
|||||||||
| Drug Name | Tramadol | Ondansetron | |||||||
| Mechanism 1 |
Serotonergic effects Serotonin transporter Inhibitor |
Serotonergic effects 5-HT 3 receptor Antagonist |
|||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Serotonin transporter |
×
Structure
Sequence
METTPLNSQKQLSACEDGEDCQENGVLQKVVPTPGDKVESGQISNGYSAVPSPGAGDDTRHSIPATTTTLVAELHQGERETWGKKVDFLLSVIGYAVDLGNVWRFPYICYQNGGGAFLLPYTIMAIFGGIPLFYMELALGQYHRNGCISIWRKICPIFKGIGYAICIIAFYIASYYNTIMAWALYYLISSFTDQLPWTSCKNSWNTGNCTNYFSEDNITWTLHSTSPAEEFYTRHVLQIHRSKGLQDLGGISWQLALCIMLIFTVIYFSIWKGVKTSGKVVWVTATFPYIILSVLLVRGATLPGAWRGVLFYLKPNWQKLLETGVWIDAAAQIFFSLGPGFGVLLAFASYNKFNNNCYQDALVTSVVNCMTSFVSGFVIFTVLGYMAEMRNEDVSEVAKDAGPSLLFITYAEAIANMPASTFFAIIFFLMLITLGLDSTFAGLEGVITAVLDEFPHVWAKRRERFVLAVVITCFFGSLVTLTFGGAYVVKLLEEYATGPAVLTVALIEAVAVSWFYGITQFCRDVKEMLGFSPGWFWRICWVAISPLFLLFIICSFLMSPPQLRLFQYNYPYWSIILGYCIGTSSFICIPTYIAYRLIITPGTFKERIIKSITPETPTEIPCGDIRLNAV
|
|||||||
| Gene Name | SLC6A4 | ||||||||
| Uniprot ID | SC6A4_HUMAN | ||||||||
| KEGG Pathway | hsa:6532 | ||||||||
| Protein Family | Sodium:neurotransmitter symporter (SNF) (TC 2.A.22) family | ||||||||
| Protein Function |
Serotonin transporter whose primary function in the central nervous system involves the regulation of serotonergic signaling via transport of serotonin molecules from the synaptic cleft back into the pre-synaptic terminal for re-utilization. Plays a key role in mediating regulation of the availability of serotonin to other receptors of serotonergic systems. Terminates the action of serotonin and recycles it in a sodium-dependent manner.
Click to Show/Hide
|
||||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | 5-HT 3 receptor | Structure Sequence | |||||||
| Protein Family | Ligand-gated ion channel (TC 1.A.9) family | ||||||||
| Protein Function |
This is one of the several different receptors for 5-hydroxytryptamine (serotonin), a biogenic hormone that functions as a neurotransmitter, a hormone, and a mitogen. This receptor is a ligand-gated ion channel, which when activated causes fast, depolarizing responses in neurons. It is a cation-specific, but otherwise relatively nonselective, ion channel.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Increased risk of prolong QT interval Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Tramadol | Ondansetron | |||||||
| Mechanism 2 | Prolong QT interval | Prolong QT interval | |||||||
| Key Mechanism Factor 3 | |||||||||
| Factor Name | QT interval | ||||||||
| Factor Description | Long QT syndrome is a heart signaling disorder that can cause a fast, chaotic heartbeat (arrhythmia). Many people may not exhibit symptoms, and usually the condition is detected during routine medical tests. In others, the most common symptoms include: sudden fainting, palpitations, dizziness, seizures, sudden death. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Coadministration of 5-HT3 receptor antagonists with tramadol should generally be avoided. Patients should be closely monitored for symptoms of the serotonin syndrome during treatment. Particular caution is warranted when initiating or increasing the dosages of these agents. In addition, the potential risk for serotonin syndrome should be considered even when administering serotonergic agents sequentially, as some agents may demonstrate a prolonged elimination half-life. If serotonin syndrome develops or is suspected during the course of therapy, all serotonergic agents should be discontinued immediately and supportive care rendered as necessary. Moderately ill patients may benefit from the administration of a serotonin antagonist (e.g., cyproheptadine, chlorpromazine). Severe cases should be managed under consultation with a toxicologist and may require sedation, neuromuscular paralysis, intubation, and mechanical ventilation in addition to the other measures. The possibility of a diminished therapeutic response to tramadol should also be considered during concomitant therapy with 5-HT3 receptor antagonists. Due to the potential for additive effects on the QT interval, ECG monitoring may also be appropriate. Particular care should be exercised in patients suspected to be at an increased risk of torsade de pointes. Patients should be advised to seek prompt medical attention if they experience symptoms that could indicate the occurrence of torsade de pointes such as dizziness, lightheadedness, fainting, palpitation, irregular heart rhythm, shortness of breath, or syncope. | ||||||||
