Details of Drug-Drug Interaction
| Drug General Information (ID: DDI7UPCIM0) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Olsalazine | Drug Info | Human cytomegalovirus immune globulin | Drug Info | |||||
| Drug Type | Small molecule | Polyclonal antibody | |||||||
| Therapeutic Class | Immunosuppressive Agents | Immune Globulins | |||||||
| Mechanism of Olsalazine-Human cytomegalovirus immune globulin Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
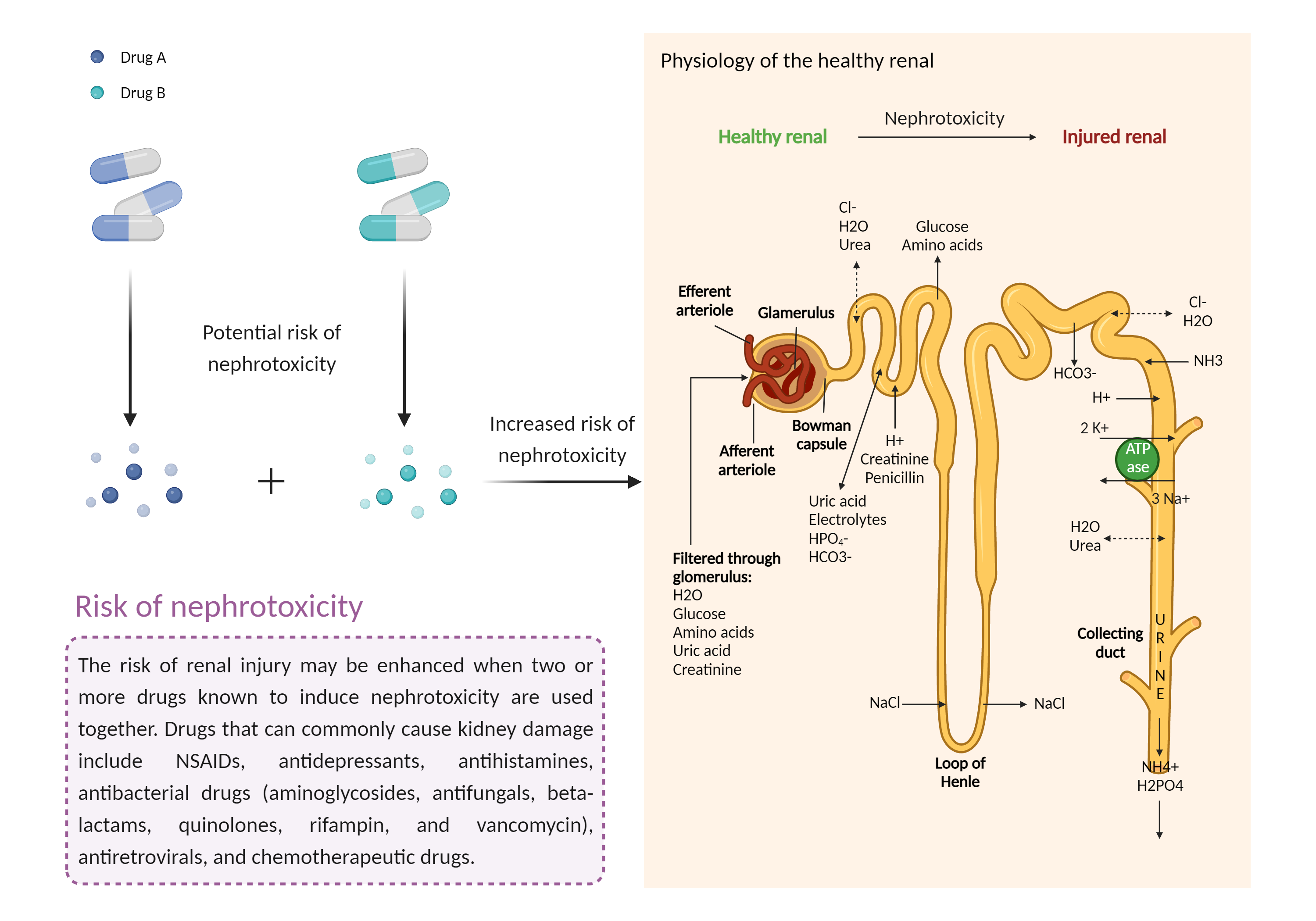
| Increased risk of nephrotoxicity Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Olsalazine | Human cytomegalovirus immune globulin | |||||||
| Mechanism | Nephrotoxicity | Nephrotoxicity | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Nephrotoxicity | ||||||||
| Factor Description | The combination of drugs that can induce nephrotoxicity may increase the risk of kidney injury. When kidney injury occurs, the inability to remove excess urine and waste from the body can lead to high levels of urea nitrogen, creatinine, and electrolytes (such as potassium and magnesium) in the blood. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Intravenous immune globulin preparations should be administered cautiously in patients treated with other potentially nephrotoxic agents (e.g., e.g., aminoglycosides polypeptide, glycopeptide, and polymyxin antibiotics amphotericin B adefovir cidofovir tenofovir foscarnet cisplatin deferasirox gallium nitrate lithium mesalamine certain immunosuppressants intravenous bisphosphonates intravenous pentamidine high intravenous dosages of methotrexate high dosages and/or chronic use of nonsteroidal anti-inflammatory agents). Clinicians should ensure that patients are not volume depleted prior to the initiation of immune globulin therapy. Monitoring of urine output and renal function tests, including the measurement of blood urea nitrogen (BUN) and serum creatinine, is recommended prior to the initial infusion and at appropriate intervals thereafter. | ||||||||
