Details of Drug-Drug Interaction
| Drug General Information (ID: DDI7MF6JI9) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Nitisinone | Drug Info | Sitagliptin | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antineoplastics | Antidiabetic Agents | |||||||
| Structure | |||||||||
| Mechanism of Nitisinone-Sitagliptin Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Transporter inhibition Click to Show/Hide Mechanism Graph | |||||||||
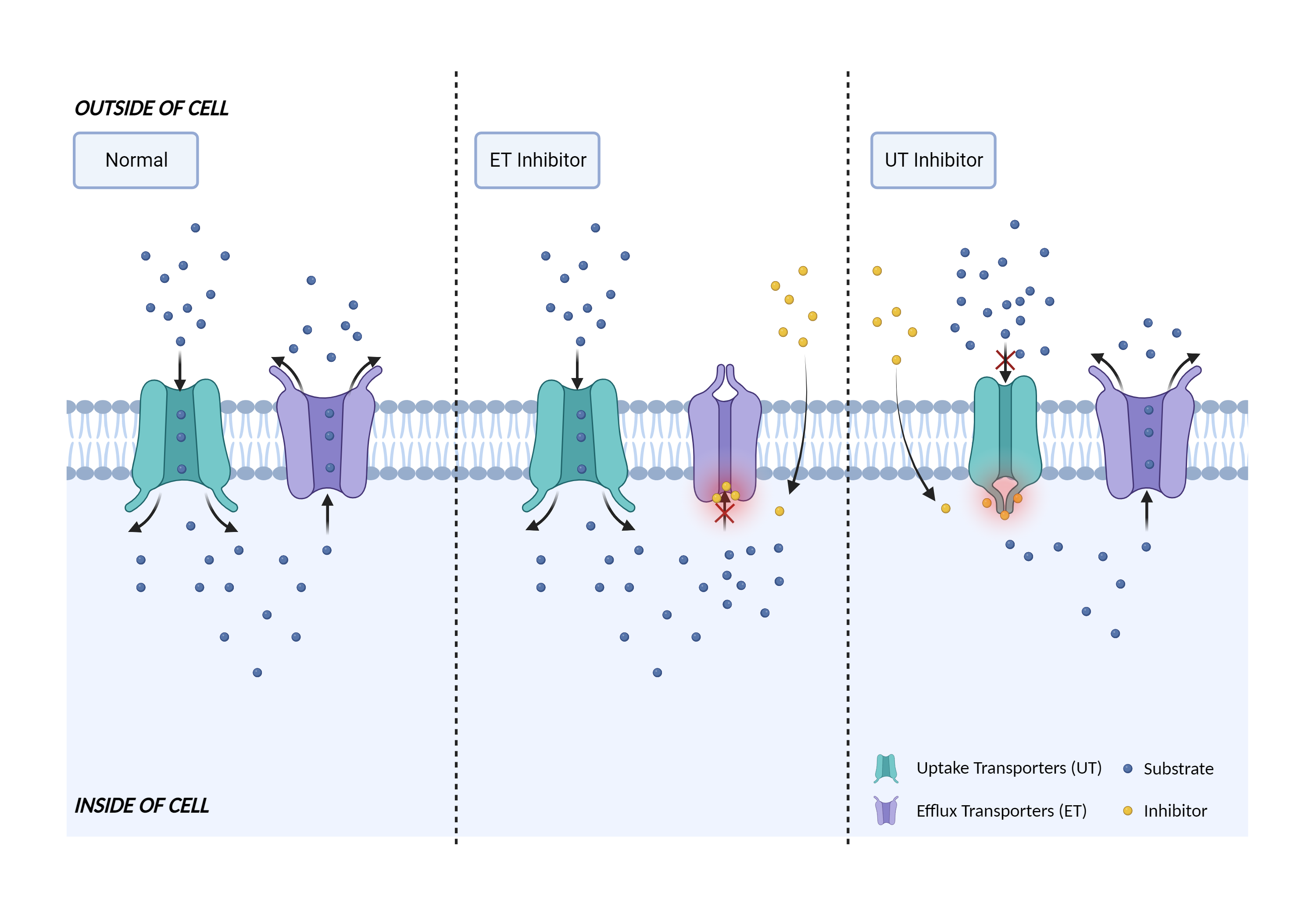 |
|||||||||
| Drug Name | Nitisinone | Sitagliptin | |||||||
| Mechanism | OAT3 inhibitor | OAT3 substrate | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Solute carrier family 22 member 8 |
×
Structure
Sequence
MTFSEILDRVGSMGHFQFLHVAILGLPILNMANHNLLQIFTAATPVHHCRPPHNASTGPWVLPMGPNGKPERCLRFVHPPNASLPNDTQRAMEPCLDGWVYNSTKDSIVTEWDLVCNSNKLKEMAQSIFMAGILIGGLVLGDLSDRFGRRPILTCSYLLLAASGSGAAFSPTFPIYMVFRFLCGFGISGITLSTVILNVEWVPTRMRAIMSTALGYCYTFGQFILPGLAYAIPQWRWLQLTVSIPFFVFFLSSWWTPESIRWLVLSGKSSKALKILRRVAVFNGKKEEGERLSLEELKLNLQKEISLAKAKYTASDLFRIPMLRRMTFCLSLAWFATGFAYYSLAMGVEEFGVNLYILQIIFGGVDVPAKFITILSLSYLGRHTTQAAALLLAGGAILALTFVPLDLQTVRTVLAVFGKGCLSSSFSCLFLYTSELYPTVIRQTGMGVSNLWTRVGSMVSPLVKITGEVQPFIPNIIYGITALLGGSAALFLPETLNQPLPETIEDLENWSLRAKKPKQEPEVEKASQRIPLQPHGPGLGSS
|
|||||||
| Gene Name | OAT3 | ||||||||
| Uniprot ID | S22A8_HUMAN | ||||||||
| KEGG Pathway | hsa:9376 | ||||||||
| Protein Family | Major facilitator (TC 2.A.1) superfamily | ||||||||
| Protein Function |
Plays an important role in the excretion/detoxification of endogenous and exogenous organic anions, especially from the brain and kidney. Involved in the transport basolateral of steviol, fexofenadine. Transports benzylpenicillin (PCG), estrone-3-sulfate (E1S), cimetidine (CMD), 2,4-dichloro-phenoxyacetate (2,4-D), p-amino-hippurate (PAH), acyclovir (ACV) and ochratoxin (OTA).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised if nitisinone is used in combination with OAT1/OAT3 substrates. Patients should be monitored for potential adverse reactions related to increased plasma concentrations of the OAT1/OAT3 substrate. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Orfadin (nitisinone). Orphan Pharmaceuticals USA, Inc, Nashville, TN. | ||||||||||||||||||
