Details of Drug-Drug Interaction
| Drug General Information (ID: DDI79Y4GAT) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Thalidomide | Drug Info | Ambenonium | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Immunosuppressive Agents | Antiarrhythmic Agents | |||||||
| Structure | |||||||||
| Mechanism of Thalidomide-Ambenonium Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
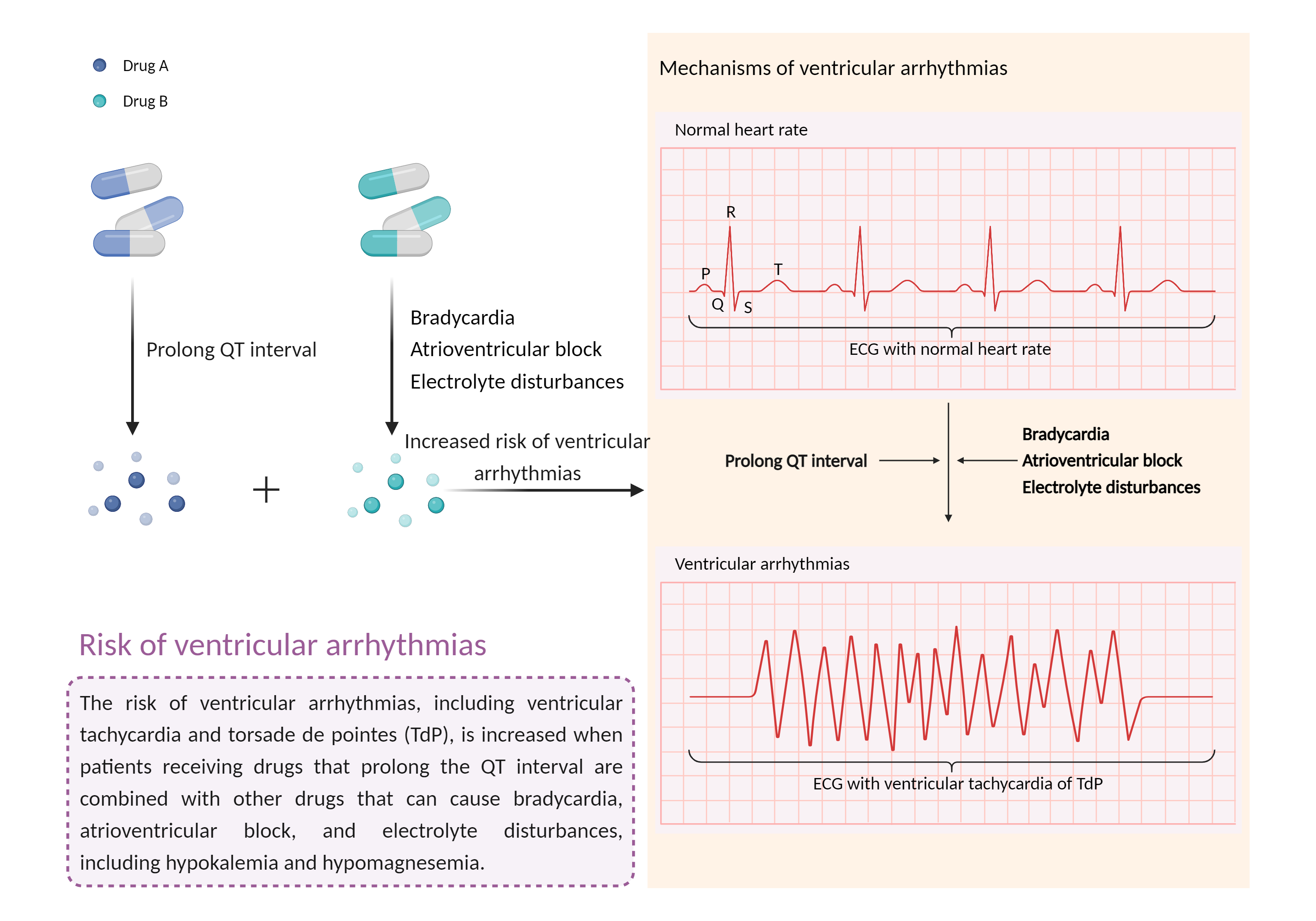
| Increased risk of ventricular arrhythmias Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Thalidomide | Ambenonium | |||||||
| Mechanism | Bradycardia | Prolong QT interval | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Ventricular arrhythmias | ||||||||
| Factor Description | Ventricular arrhythmias are abnormal heart rhythms that cause your heart's lower chambers to pump blood instead of pumping it. This can limit or stop your heart from supplying blood to your body. While some of these arrhythmias are harmless and do not cause symptoms, others can have serious, even fatal, effects on your body. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised when thalidomide is used with drugs that can slow the heart rate or induce torsade de pointes arrhythmia. Patients should be monitored for bradycardia, atrioventricular block and syncope, and advised to seek medical attention if they experience dizziness, lightheadedness, fainting, shortness of breath, or slow or irregular heartbeat. A dose reduction of thalidomide or discontinuation may be required. | ||||||||
