Details of Drug-Drug Interaction
| Drug General Information (ID: DDI74JCLNQ) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Delavirdine | Drug Info | Terbinafine | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Anti-Hiv Agents | Antifungal Agents | |||||||
| Structure | |||||||||
| Mechanism of Delavirdine-Terbinafine Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| CYP450 enzyme inhibition Click to Show/Hide Mechanism Graph | |||||||||
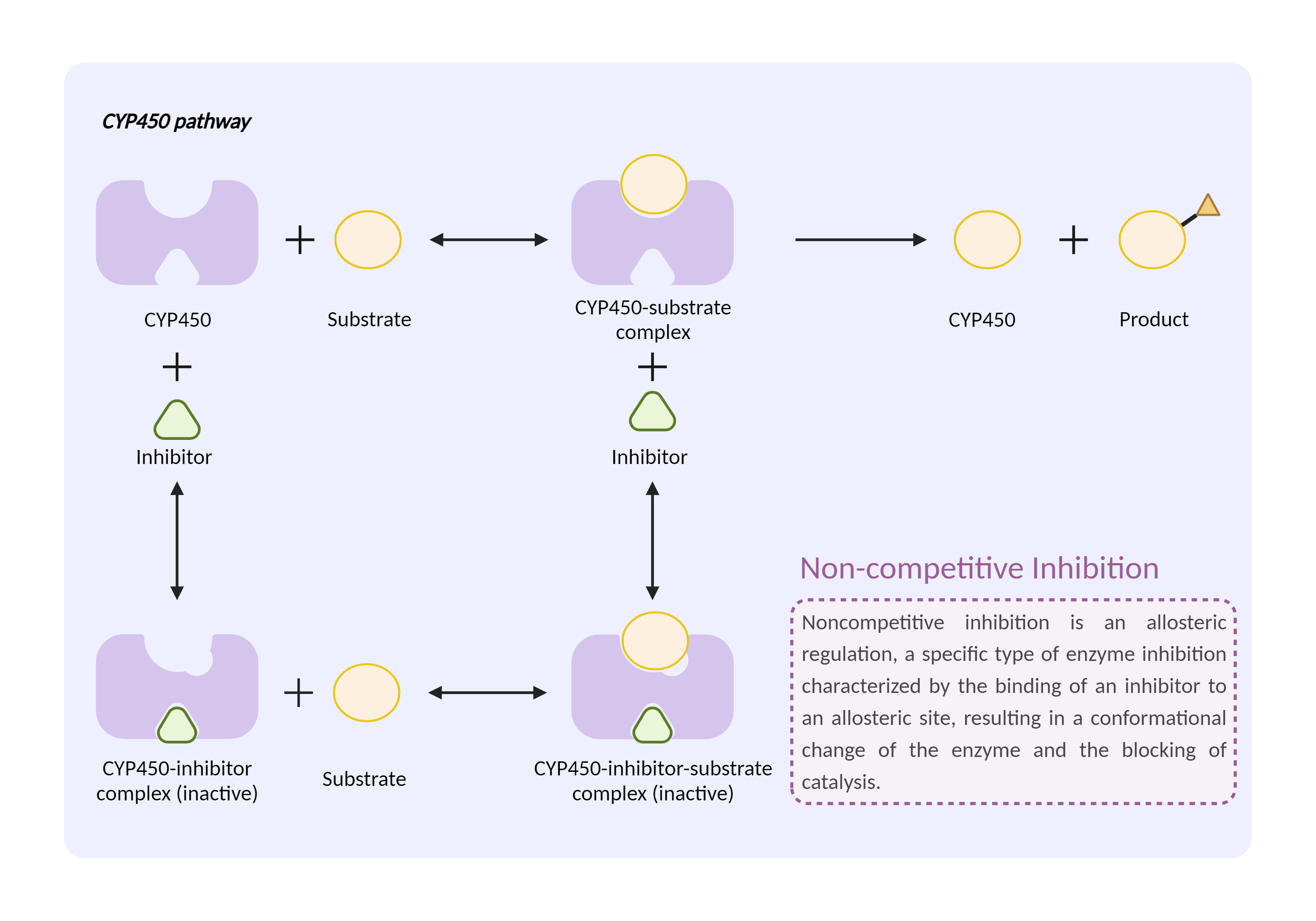 |
|||||||||
| Drug Name | Delavirdine | Terbinafine | |||||||
| Mechanism | CYP450 2D6 substrate | CYP450 2D6 inhibitor | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Cytochrome P450 2D6 |
×
Structure
Sequence
MGLEALVPLAVIVAIFLLLVDLMHRRQRWAARYPPGPLPLPGLGNLLHVDFQNTPYCFDQLRRRFGDVFSLQLAWTPVVVLNGLAAVREALVTHGEDTADRPPVPITQILGFGPRSQGVFLARYGPAWREQRRFSVSTLRNLGLGKKSLEQWVTEEAACLCAAFANHSGRPFRPNGLLDKAVSNVIASLTCGRRFEYDDPRFLRLLDLAQEGLKEESGFLREVLNAVPVLLHIPALAGKVLRFQKAFLTQLDELLTEHRMTWDPAQPPRDLTEAFLAEMEKAKGNPESSFNDENLRIVVADLFSAGMVTTSTTLAWGLLLMILHPDVQRRVQQEIDDVIGQVRRPEMGDQAHMPYTTAVIHEVQRFGDIVPLGVTHMTSRDIEVQGFRIPKGTTLITNLSSVLKDEAVWEKPFRFHPEHFLDAQGHFVKPEAFLPFSAGRRACLGEPLARMELFLFFTSLLQHFSFSVPTGQPRPSHHGVFAFLVSPSPYELCAVPR
|
|||||||
| Gene Name | CYP2D6 | ||||||||
| Uniprot ID | CP2D6_HUMAN | ||||||||
| KEGG Pathway | hsa:1565 | ||||||||
| Protein Family | Cytochrome P450 family | ||||||||
| Protein Function |
A cytochrome P450 monooxygenase involved in the metabolism of fatty acids, steroids and retinoids (PubMed:18698000, PubMed:19965576, PubMed:20972997, PubMed:21289075, PubMed:21576599). Mechanistically, uses molecular oxygen inserting one oxygen atom into a substrate, and reducing the second into a water molecule, with two electrons provided by NADPH via cytochrome P450 reductase (NADPH--hemoprotein reductase) (PubMed:18698000, PubMed:19965576, PubMed:20972997, PubMed:21289075, PubMed:21576599). Catalyzes the epoxidation of double bonds of polyunsaturated fatty acids (PUFA) (PubMed:19965576, PubMed:20972997). Metabolizes endocannabinoid arachidonoylethanolamide (anandamide) to 20-hydroxyeicosatetraenoic acid ethanolamide (20-HETE-EA) and 8,9-, 11,12-, and 14,15-epoxyeicosatrienoic acid ethanolamides (EpETrE-EAs), potentially modulating endocannabinoid system signaling (PubMed:18698000, PubMed:21289075). Catalyzes the hydroxylation of carbon-hydrogen bonds. Metabolizes cholesterol toward 25-hydroxycholesterol, a physiological regulator of cellular cholesterol homeostasis (PubMed:21576599). Catalyzes the oxidative transformations of all-trans retinol to all-trans retinal, a precursor for the active form all-trans-retinoic acid (PubMed:10681376). Also involved in the oxidative metabolism of drugs such as antiarrhythmics, adrenoceptor antagonists, and tricyclic antidepressants.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised if terbinafine must be used concurrently with medications that undergo metabolism by CYP450 2D6, particularly those with a narrow therapeutic range. Dosage adjustments as well as clinical and laboratory monitoring may be appropriate for some drugs whenever terbinafine is added to or withdrawn from therapy. Due to the long elimination half-life of terbinafine, especially following prolonged use, such interactions may be observed for several months after discontinuation of terbinafine therapy. | ||||||||
