Details of Drug-Drug Interaction
| Drug General Information (ID: DDI6NYE2ZL) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Natalizumab | Drug Info | Talazoparib | Drug Info | |||||
| Drug Type | Monoclonal antibody | Small molecule | |||||||
| Therapeutic Class | Selective Immunosuppressants | Poly (Adp-Ribose) Polymerase Inhibitors | |||||||
| Mechanism of Natalizumab-Talazoparib Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Additive immunosuppressive effects Click to Show/Hide Mechanism Graph | |||||||||
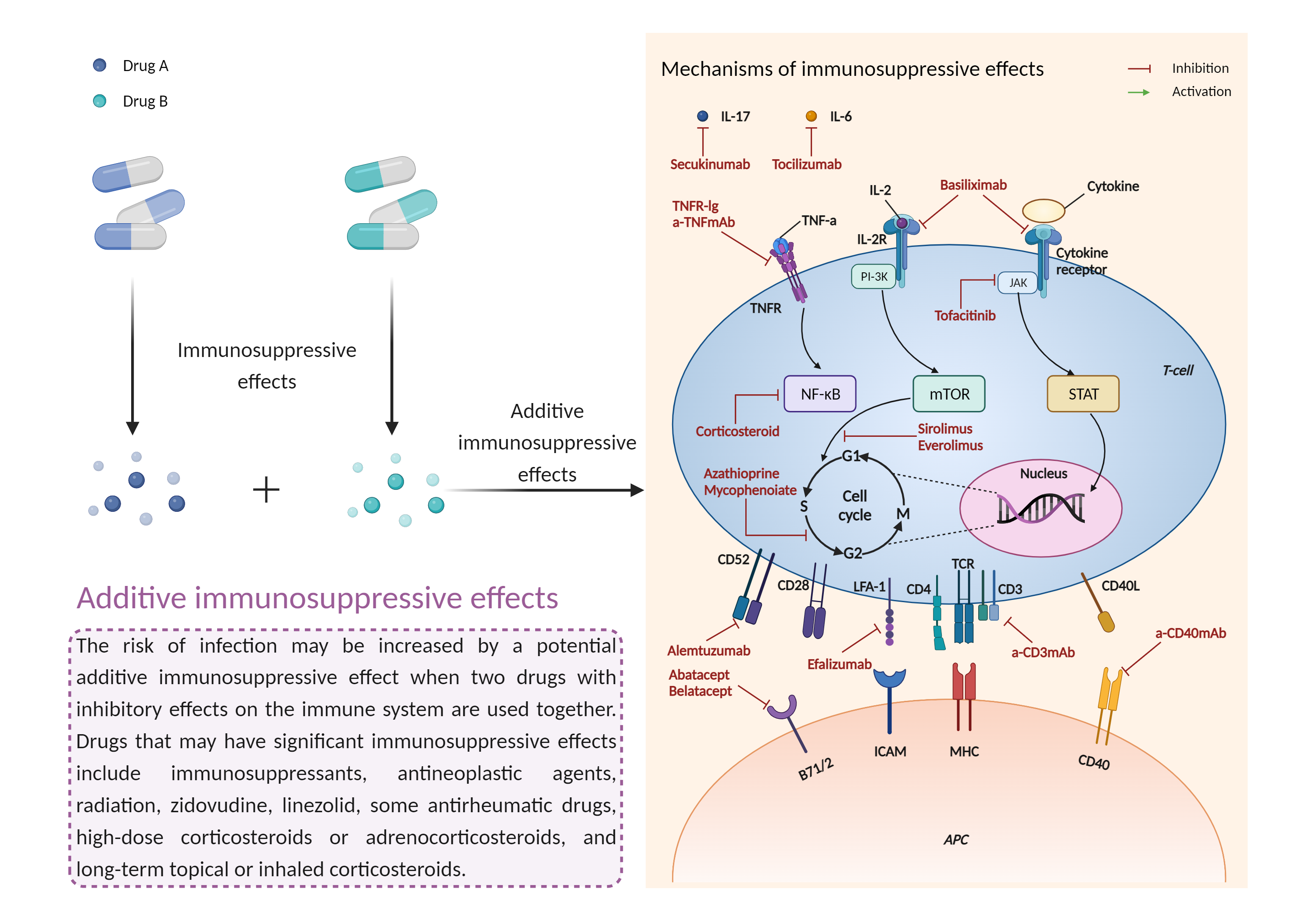 |
|||||||||
| Drug Name | Natalizumab | Talazoparib | |||||||
| Mechanism | Immunosuppressive effects | Immunosuppressive effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Immunosuppressive effects | ||||||||
| Factor Description | Immunosuppression is when your immune system is not functioning as it should. The immune system is made up of cells, tissues and organs that help the body fight off infections. If the immune system is suppressed, an infection that your body was able to control may become serious or even fatal. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | The safety and efficacy of natalizumab in combination with immunosuppressant, immunomodulating, antineoplastic or other myelosuppressive agents have not been established. In general, patients receiving chronic therapy with such agents should not be treated with natalizumab due to potentially increased risk of PML and other serious infections. Typical symptoms associated with PML are diverse, progress over days to weeks, and include progressive weakness on one side of the body, clumsiness of limbs, disturbance of vision, and changes in thinking, memory, and orientation leading to confusion and personality changes. Due to the long half-life of natalizumab, immune effects are possible for up to 2 to 3 months following its discontinuation. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Tysabri (natalizumab). Elan Pharmaceutical/Athena Neurosciences Inc, South San Francisco, CA. | ||||||||||||||||||
