Details of Drug-Drug Interaction
| Drug General Information (ID: DDI6E2LHMJ) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Brentuximab vedotin | Drug Info | Regorafenib | Drug Info | |||||
| Drug Type | Monoclonal antibody | Small molecule | |||||||
| Therapeutic Class | Antineoplastics | Vegf/Vegfr Inhibitors | |||||||
| Mechanism of Brentuximab vedotin-Regorafenib Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
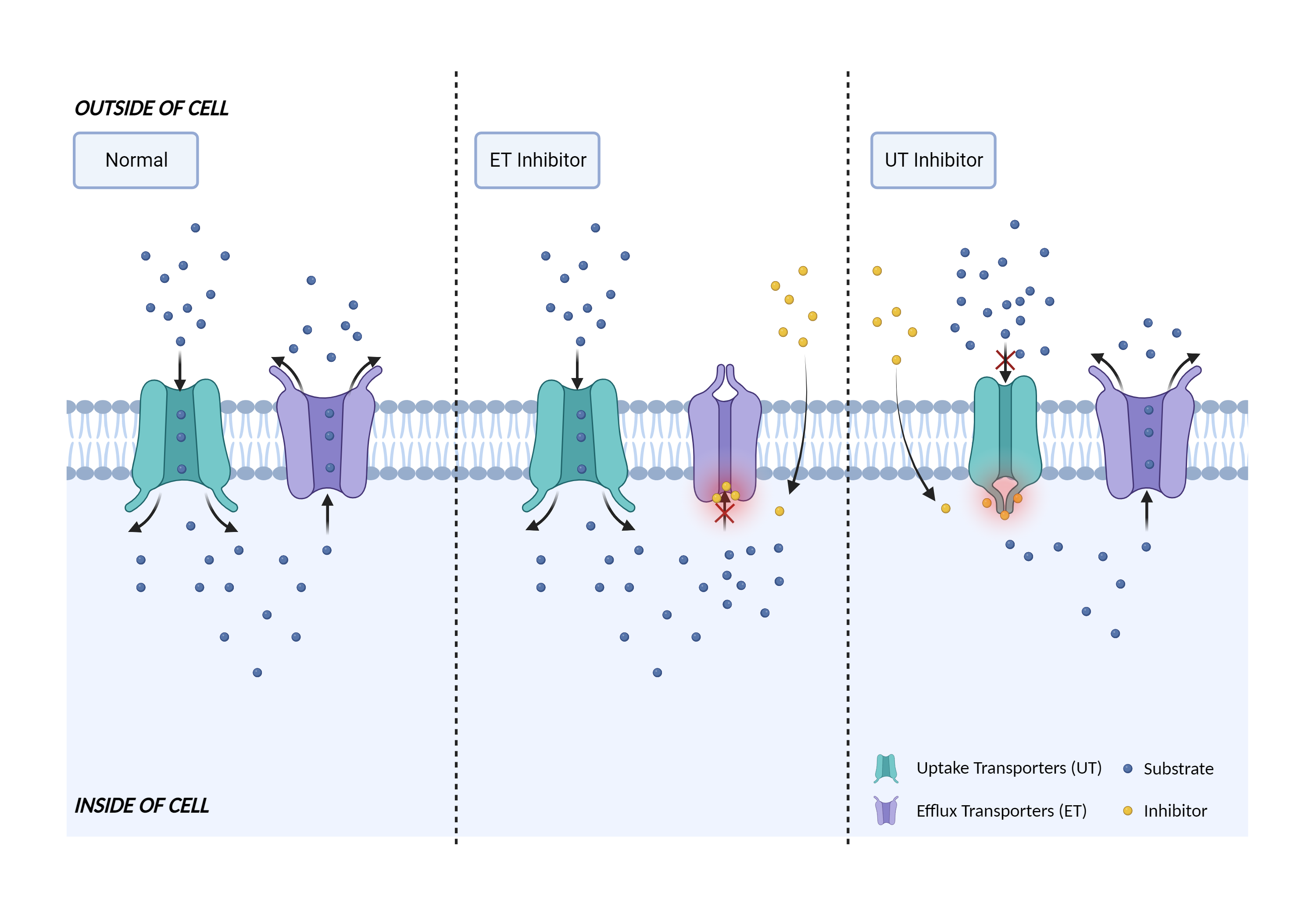
| Transporter inhibition Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Brentuximab vedotin | Regorafenib | |||||||
| Mechanism 1 | P-gp substrate | P-gp inhibitor | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | P-glycoprotein 1 |
×
Structure
Sequence
MDLEGDRNGGAKKKNFFKLNNKSEKDKKEKKPTVSVFSMFRYSNWLDKLYMVVGTLAAIIHGAGLPLMMLVFGEMTDIFANAGNLEDLMSNITNRSDINDTGFFMNLEEDMTRYAYYYSGIGAGVLVAAYIQVSFWCLAAGRQIHKIRKQFFHAIMRQEIGWFDVHDVGELNTRLTDDVSKINEGIGDKIGMFFQSMATFFTGFIVGFTRGWKLTLVILAISPVLGLSAAVWAKILSSFTDKELLAYAKAGAVAEEVLAAIRTVIAFGGQKKELERYNKNLEEAKRIGIKKAITANISIGAAFLLIYASYALAFWYGTTLVLSGEYSIGQVLTVFFSVLIGAFSVGQASPSIEAFANARGAAYEIFKIIDNKPSIDSYSKSGHKPDNIKGNLEFRNVHFSYPSRKEVKILKGLNLKVQSGQTVALVGNSGCGKSTTVQLMQRLYDPTEGMVSVDGQDIRTINVRFLREIIGVVSQEPVLFATTIAENIRYGRENVTMDEIEKAVKEANAYDFIMKLPHKFDTLVGERGAQLSGGQKQRIAIARALVRNPKILLLDEATSALDTESEAVVQVALDKARKGRTTIVIAHRLSTVRNADVIAGFDDGVIVEKGNHDELMKEKGIYFKLVTMQTAGNEVELENAADESKSEIDALEMSSNDSRSSLIRKRSTRRSVRGSQAQDRKLSTKEALDESIPPVSFWRIMKLNLTEWPYFVVGVFCAIINGGLQPAFAIIFSKIIGVFTRIDDPETKRQNSNLFSLLFLALGIISFITFFLQGFTFGKAGEILTKRLRYMVFRSMLRQDVSWFDDPKNTTGALTTRLANDAAQVKGAIGSRLAVITQNIANLGTGIIISFIYGWQLTLLLLAIVPIIAIAGVVEMKMLSGQALKDKKELEGSGKIATEAIENFRTVVSLTQEQKFEHMYAQSLQVPYRNSLRKAHIFGITFSFTQAMMYFSYAGCFRFGAYLVAHKLMSFEDVLLVFSAVVFGAMAVGQVSSFAPDYAKAKISAAHIIMIIEKTPLIDSYSTEGLMPNTLEGNVTFGEVVFNYPTRPDIPVLQGLSLEVKKGQTLALVGSSGCGKSTVVQLLERFYDPLAGKVLLDGKEIKRLNVQWLRAHLGIVSQEPILFDCSIAENIAYGDNSRVVSQEEIVRAAKEANIHAFIESLPNKYSTKVGDKGTQLSGGQKQRIAIARALVRQPHILLLDEATSALDTESEKVVQEALDKAREGRTCIVIAHRLSTIQNADLIVVFQNGRVKEHGTHQQLLAQKGIYFSMVSVQAGTKRQ
|
|||||||
| Gene Name | ABCB1 | ||||||||
| Uniprot ID | MDR1_HUMAN | ||||||||
| KEGG Pathway | hsa:5243 | ||||||||
| Protein Family | ABC transporter superfamily | ||||||||
| Protein Function |
Translocates drugs and phospholipids across the membrane (PubMed:8898203, PubMed:2897240, PubMed:9038218). Catalyzes the flop of phospholipids from the cytoplasmic to the exoplasmic leaflet of the apical membrane. Participates mainly to the flop of phosphatidylcholine, phosphatidylethanolamine, beta-D-glucosylceramides and sphingomyelins (PubMed:8898203). Energy-dependent efflux pump responsible for decreased drug accumulation in multidrug-resistant cells (PubMed:2897240, PubMed:9038218).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
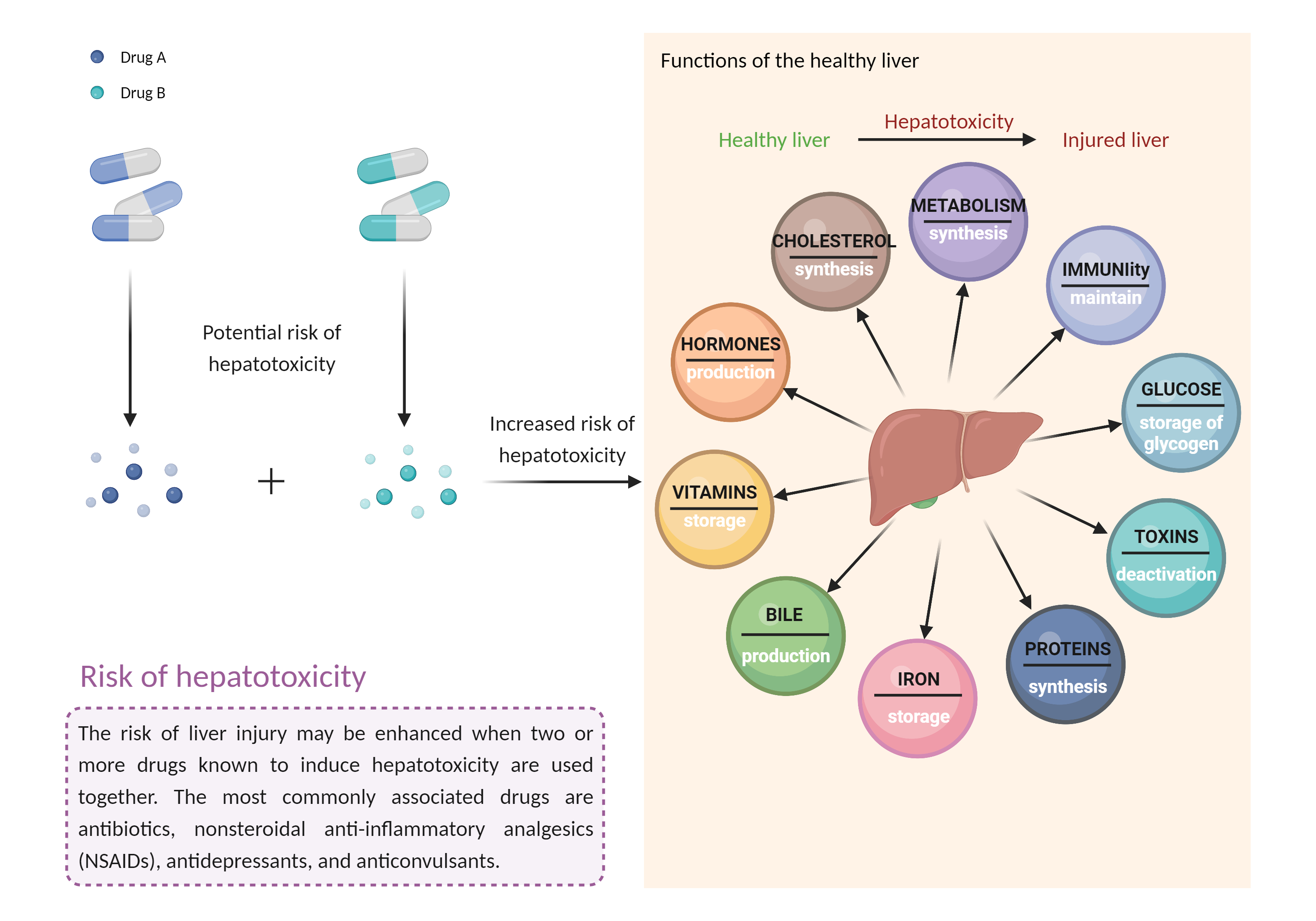
| Increased risk of hepatotoxicity Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Brentuximab vedotin | Regorafenib | |||||||
| Mechanism 2 | Hepatotoxicity | Hepatotoxicity | |||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Hepatotoxicity | ||||||||
| Factor Description | Combination of drugs that can induce hepatotoxicity may increase the risk of liver injury. Symptoms vary depending on the level of exposure and the total extent of liver damage, and may cause few symptoms if the damage is mild, and eventually lead to liver failure in patients with severe damage. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised when brentuximab is used with potent CYP450 3A4 inhibitors (e.g., azole antifungal agents, clarithromycin, erythromycin, nefazodone, ritonavir, telithromycin) or P-gp inhibitors (e.g., protein kinase inhibitors, abiraterone, amiodarone, azithromycin, cyclosporine, dronedarone, ivacaftor) that are also potentially hepatotoxic. Close monitoring for adverse effects including neutropenia, infection, peripheral neuropathy, and hepatotoxicity is recommended. Patients should be advised to seek medical attention if they experience potential signs and symptoms of hepatotoxicity such as fever, rash, itching, anorexia, nausea, vomiting, fatigue, malaise, right upper quadrant pain, dark urine, pale stools, and jaundice. Liver enzymes and bilirubin should be measured before and during treatment, especially in patients with underlying hepatic disease or marked baseline transaminase elevations. | ||||||||
