Details of Drug-Drug Interaction
| Drug General Information (ID: DDI5X7VFD2) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Repaglinide | Drug Info | Gemfibrozil | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antidiabetic Agents | Antihyperammonia Agents | |||||||
| Structure | |||||||||
| Mechanism of Repaglinide-Gemfibrozil Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
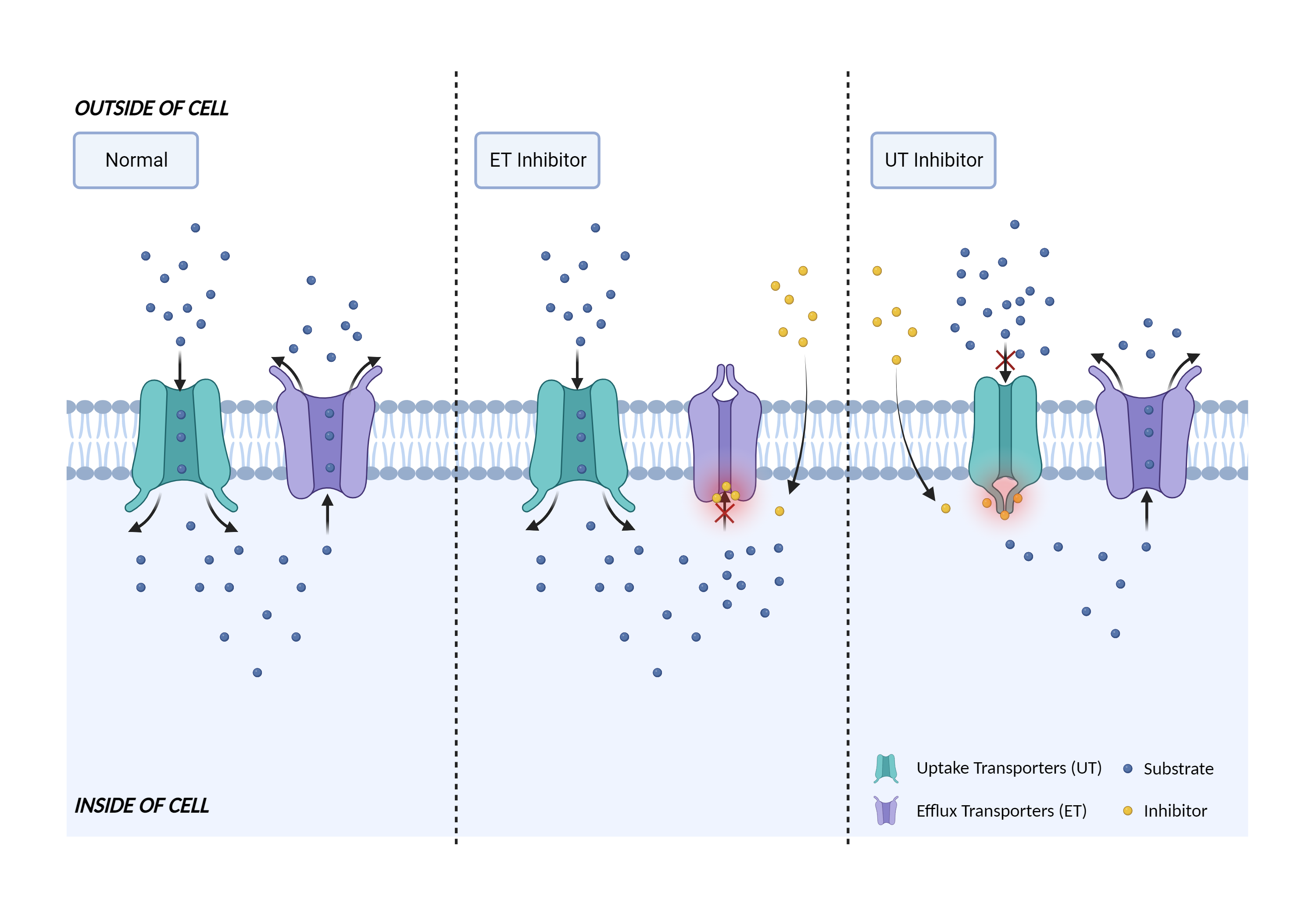
| Transporter inhibition Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Repaglinide | Gemfibrozil | |||||||
| Mechanism 1 | OATP1B1 substrate | OATP1B1 inhibitor | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Liver organic anion transporter 1 |
×
Structure
Sequence
MDQNQHLNKTAEAQPSENKKTRYCNGLKMFLAALSLSFIAKTLGAIIMKSSIIHIERRFEISSSLVGFIDGSFEIGNLLVIVFVSYFGSKLHRPKLIGIGCFIMGIGGVLTALPHFFMGYYRYSKETNINSSENSTSTLSTCLINQILSLNRASPEIVGKGCLKESGSYMWIYVFMGNMLRGIGETPIVPLGLSYIDDFAKEGHSSLYLGILNAIAMIGPIIGFTLGSLFSKMYVDIGYVDLSTIRITPTDSRWVGAWWLNFLVSGLFSIISSIPFFFLPQTPNKPQKERKASLSLHVLETNDEKDQTANLTNQGKNITKNVTGFFQSFKSILTNPLYVMFVLLTLLQVSSYIGAFTYVFKYVEQQYGQPSSKANILLGVITIPIFASGMFLGGYIIKKFKLNTVGIAKFSCFTAVMSLSFYLLYFFILCENKSVAGLTMTYDGNNPVTSHRDVPLSYCNSDCNCDESQWEPVCGNNGITYISPCLAGCKSSSGNKKPIVFYNCSCLEVTGLQNRNYSAHLGECPRDDACTRKFYFFVAIQVLNLFFSALGGTSHVMLIVKIVQPELKSLALGFHSMVIRALGGILAPIYFGALIDTTCIKWSTNNCGTRGSCRTYNSTSFSRVYLGLSSMLRVSSLVLYIILIYAMKKKYQEKDINASENGSVMDEANLESLNKNKHFVPSAGADSETHC
|
|||||||
| Gene Name | OATP1B1 | ||||||||
| Uniprot ID | SO1B1_HUMAN | ||||||||
| KEGG Pathway | hsa:10599 | ||||||||
| Protein Family | Organo anion transporter (TC 2.A.60) family | ||||||||
| Protein Function |
Mediates the Na(+)-independent uptake of organic anions such as pravastatin, taurocholate, methotrexate, dehydroepiandrosterone sulfate, 17-beta-glucuronosyl estradiol, estrone sulfate, prostaglandin E2, thromboxane B2, leukotriene C3, leukotriene E4, thyroxine and triiodothyronine. Involved in the clearance of bile acids and organic anions from the liver.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
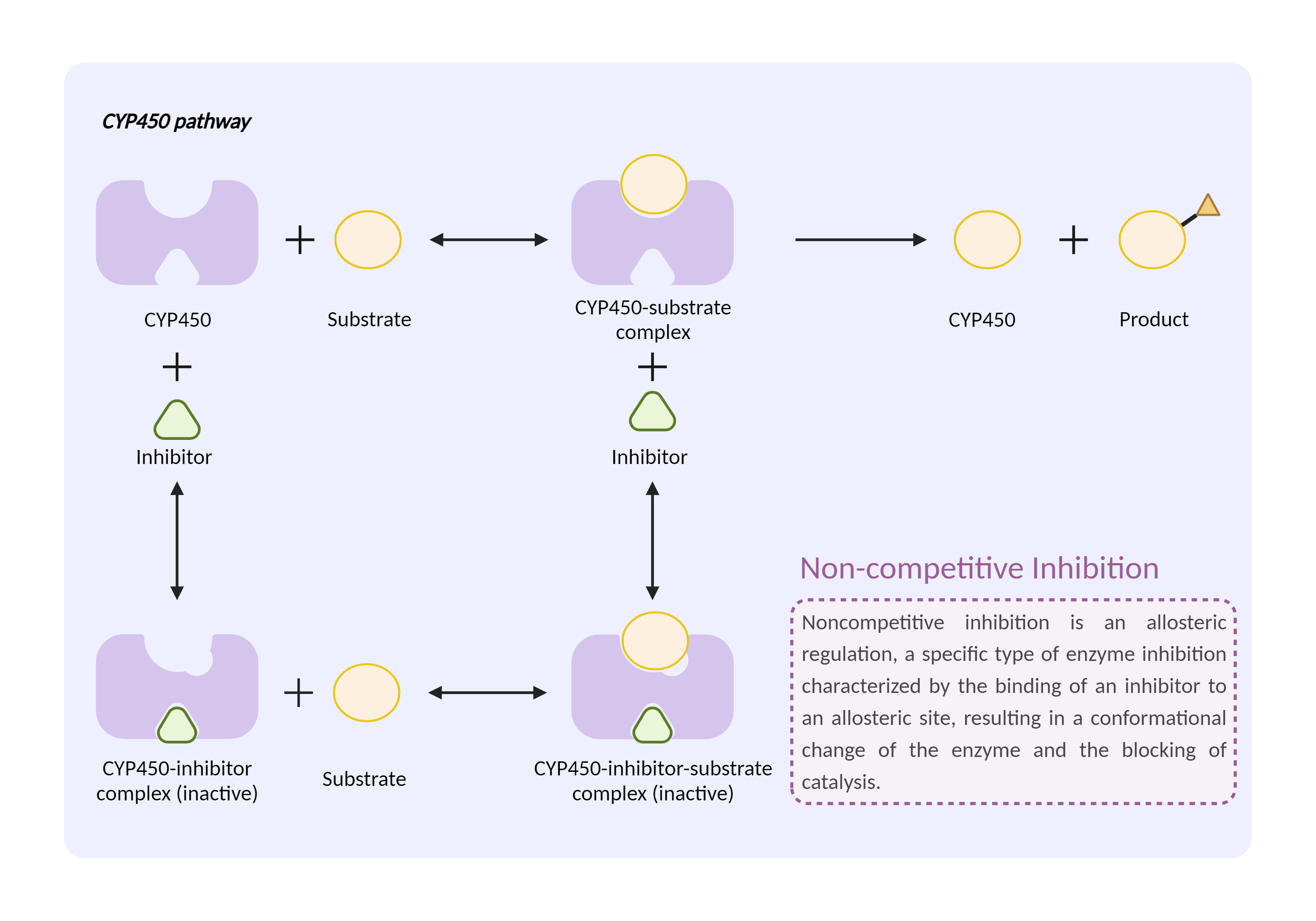
| CYP450 enzyme inhibition Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Repaglinide | Gemfibrozil | |||||||
| Mechanism 2 | CYP450 2C8 substrate | CYP450 2C8 inhibitor | |||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Cytochrome P450 2C8 |
×
Structure
Sequence
MEPFVVLVLCLSFMLLFSLWRQSCRRRKLPPGPTPLPIIGNMLQIDVKDICKSFTNFSKVYGPVFTVYFGMNPIVVFHGYEAVKEALIDNGEEFSGRGNSPISQRITKGLGIISSNGKRWKEIRRFSLTTLRNFGMGKRSIEDRVQEEAHCLVEELRKTKASPCDPTFILGCAPCNVICSVVFQKRFDYKDQNFLTLMKRFNENFRILNSPWIQVCNNFPLLIDCFPGTHNKVLKNVALTRSYIREKVKEHQASLDVNNPRDFIDCFLIKMEQEKDNQKSEFNIENLVGTVADLFVAGTETTSTTLRYGLLLLLKHPEVTAKVQEEIDHVIGRHRSPCMQDRSHMPYTDAVVHEIQRYSDLVPTGVPHAVTTDTKFRNYLIPKGTTIMALLTSVLHDDKEFPNPNIFDPGHFLDKNGNFKKSDYFMPFSAGKRICAGEGLARMELFLFLTTILQNFNLKSVDDLKNLNTTAVTKGIVSLPPSYQICFIPV
|
|||||||
| Gene Name | CYP2C8 | ||||||||
| Uniprot ID | CP2C8_HUMAN | ||||||||
| KEGG Pathway | hsa:1558 | ||||||||
| Protein Family | Cytochrome P450 family | ||||||||
| Protein Function |
A cytochrome P450 monooxygenase involved in the metabolism of various endogenous substrates, including fatty acids, steroid hormones and vitamins (PubMed:7574697, PubMed:11093772, PubMed:14559847, PubMed:15766564, PubMed:19965576). Mechanistically, uses molecular oxygen inserting one oxygen atom into a substrate, and reducing the second into a water molecule, with two electrons provided by NADPH via cytochrome P450 reductase (NADPH--hemoprotein reductase) (PubMed:7574697, PubMed:11093772, PubMed:14559847, PubMed:15766564, PubMed:19965576). Primarily catalyzes the epoxidation of double bonds of polyunsaturated fatty acids (PUFA) with a preference for the last double bond (PubMed:7574697, PubMed:15766564, PubMed:19965576). Catalyzes the hydroxylation of carbon-hydrogen bonds. Metabolizes all trans-retinoic acid toward its 4-hydroxylated form (PubMed:11093772). Displays 16-alpha hydroxylase activity toward estrogen steroid hormones, 17beta-estradiol (E2) and estrone (E1) (PubMed:14559847). Plays a role in the oxidative metabolism of xenobiotics. It is the principal enzyme responsible for the metabolism of the anti-cancer drug paclitaxel (taxol) (PubMed:26427316).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Concomitant use of repaglinide and gemfibrozil is considered contraindicated. alternatives to gemfibrozil in patients taking repaglinide include bezafibrate and fenofibrate, which have been shown not to interact pharmacokinetically with repaglinide. In patients treated with gemfibrozil with or without itraconazole, nateglinide may be an appropriate substitution for repaglinide. | ||||||||
