Details of Drug-Drug Interaction
| Drug General Information (ID: DDI5JWKCIP) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Pamidronic acid | Drug Info | Deferasirox | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antiinflammatory Agents | Chelating Agents | |||||||
| Structure | |||||||||
| Mechanism of Pamidronic acid-Deferasirox Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
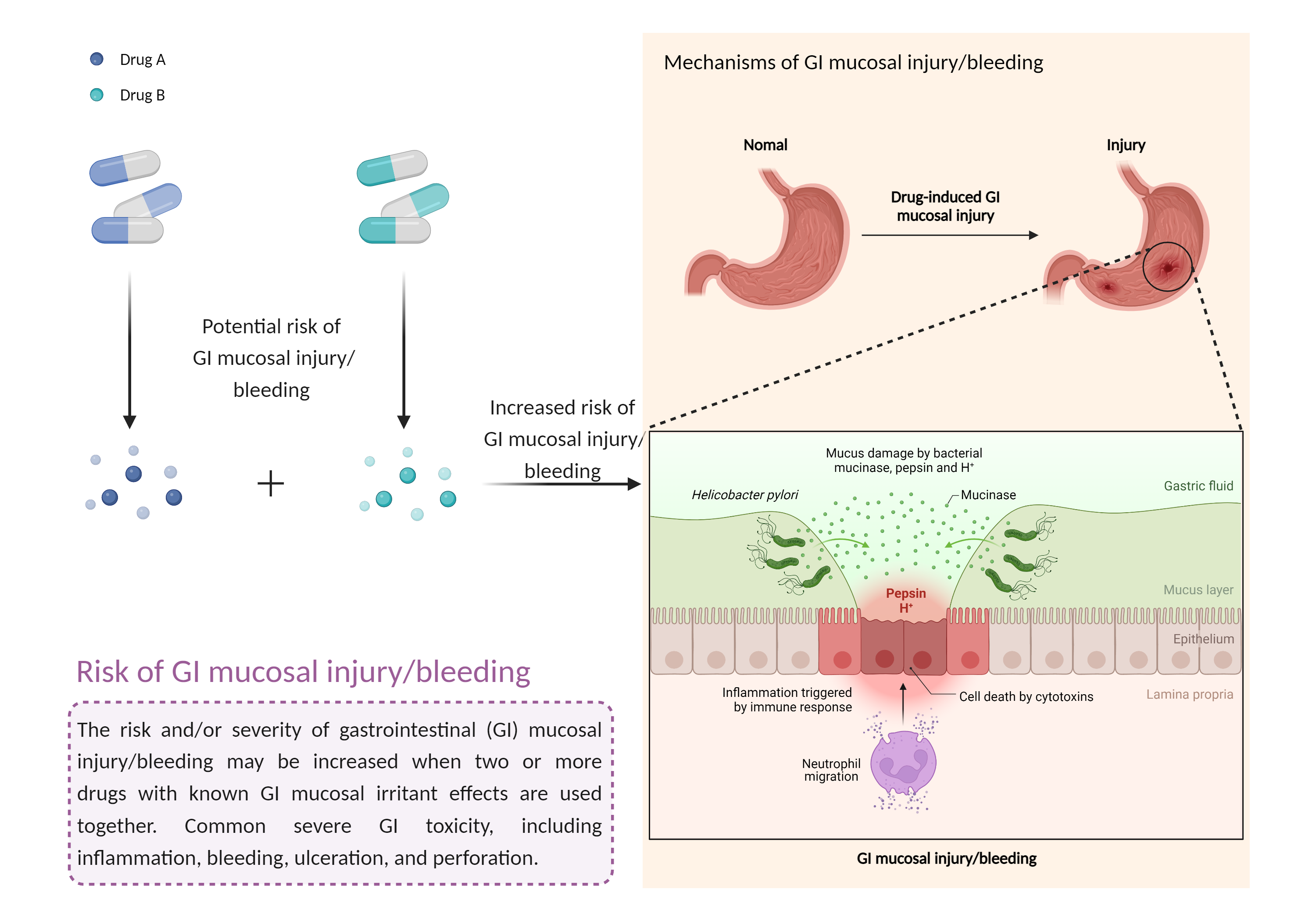
| Increased risk of GI mucosal injury/bleeding risk Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Pamidronic acid | Deferasirox | |||||||
| Mechanism 1 | GI mucosal injury/bleeding risk | GI mucosal injury/bleeding risk | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Gastrointestinal toxicity | ||||||||
| Factor Description | The risk and severity of gastrointestinal toxicity, including inflammation, bleeding, ulceration, and perforation, increases when drugs with gastrointestinal mucosal irritant effects are combined. | ||||||||
| Mechanism Description |
|
||||||||
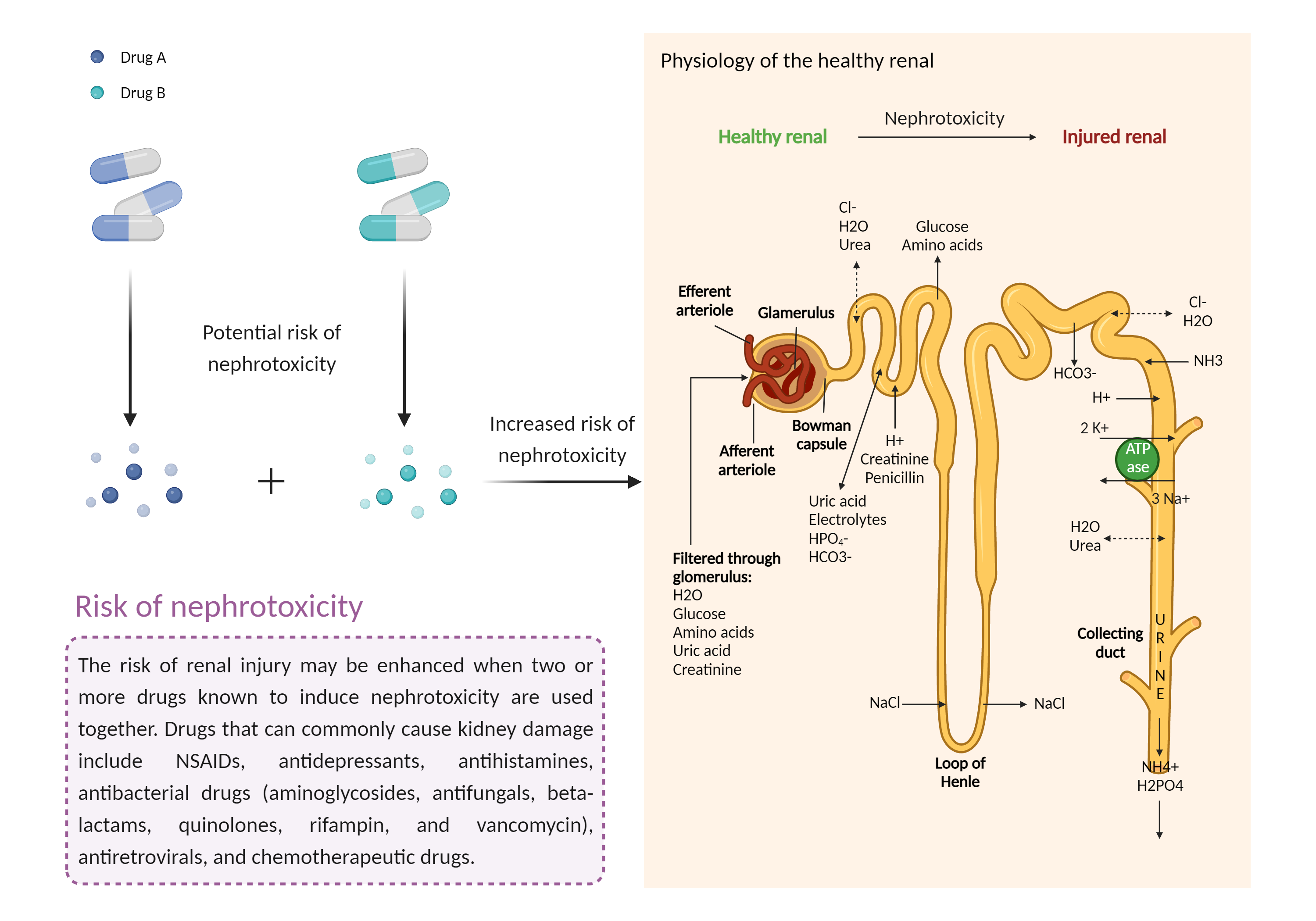
| Increased risk of nephrotoxicity Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Pamidronic acid | Deferasirox | |||||||
| Mechanism 2 | Nephrotoxicity | Nephrotoxicity | |||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Nephrotoxicity | ||||||||
| Factor Description | The combination of drugs that can induce nephrotoxicity may increase the risk of kidney injury. When kidney injury occurs, the inability to remove excess urine and waste from the body can lead to high levels of urea nitrogen, creatinine, and electrolytes (such as potassium and magnesium) in the blood. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised if deferasirox is used in combination with drugs that have known ulcerogenic potential, including oral bisphosphonates. Patients should be advised to contact their physician if they develop potential signs and symptoms of gastrointestinal injury such as abdominal pain, bloating, dizziness, lightheadedness, vomiting blood, anorexia, and/or black, tarry stools. Caution is advised if deferasirox is used in combination with other potentially nephrotoxic agents, including intravenous bisphosphonates. Serum creatinine and/or creatinine clearance should be closely monitored (e.g., prior to initiation of deferasirox therapy, then weekly during the first month after initiation or modification of therapy and monthly thereafter), especially in the elderly and patients with preexisting renal impairment, comorbid conditions, dehydration, or severe infections. Dosage reduction, interruption, or discontinuation should be considered in the presence of creatinine elevations. A progressive increase in serum creatinine beyond the age-appropriate upper limit of normal may warrant an interruption of therapy. Once the creatinine has returned to within the normal range, therapy may be reinitiated at a lower dose followed by a gradual dose escalation according to the product labeling, provided the clinical benefit is expected to outweigh potential risks. | ||||||||
