Details of Drug-Drug Interaction
| Drug General Information (ID: DDI5GCYOX3) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Teriflunomide | Drug Info | Olaparib | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Selective Immunosuppressants | Poly (Adp-Ribose) Polymerase Inhibitors | |||||||
| Structure | |||||||||
| Mechanism of Teriflunomide-Olaparib Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
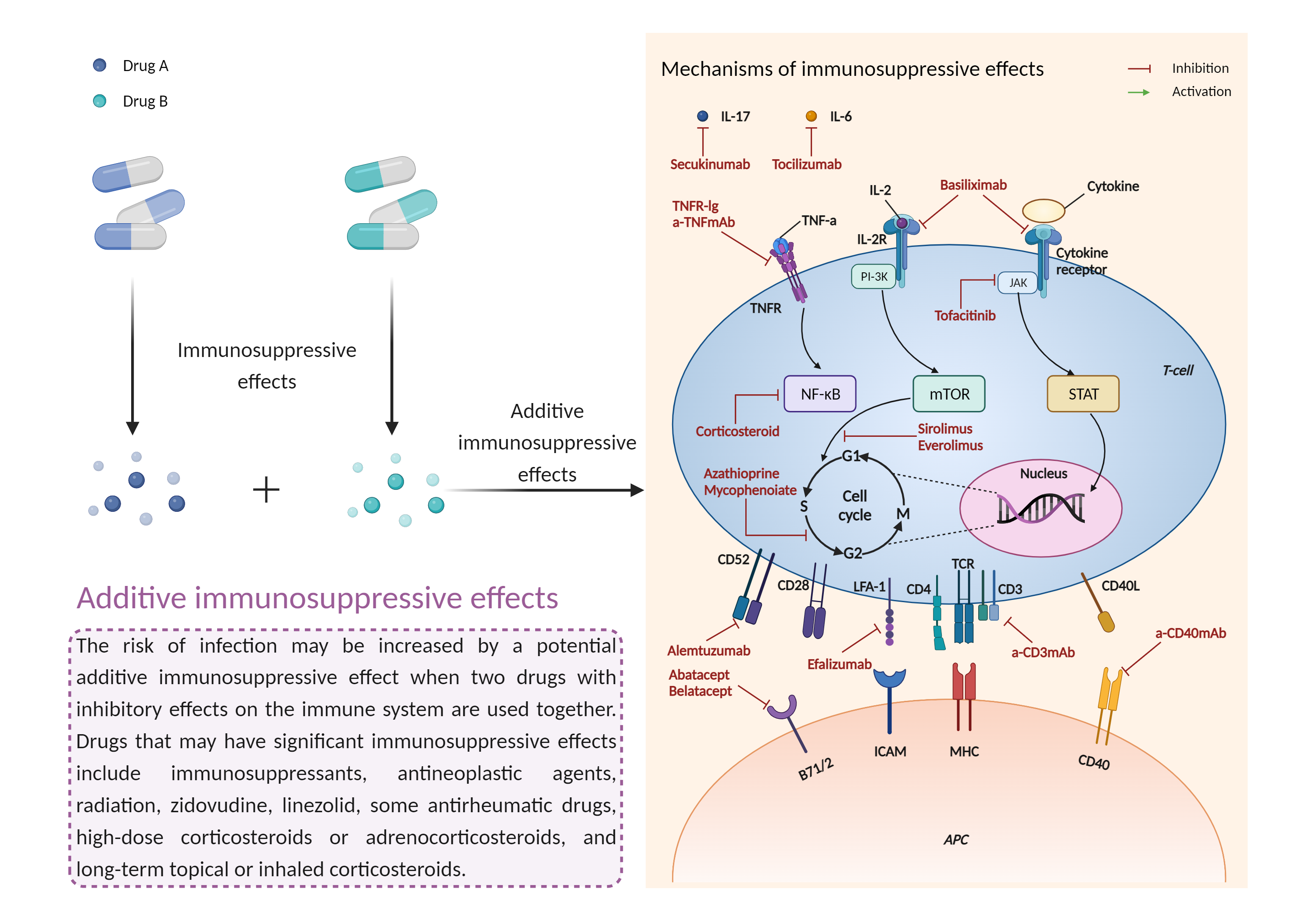
| Additive immunosuppressive effects Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Teriflunomide | Olaparib | |||||||
| Mechanism 1 | Immunosuppressive effects | Immunosuppressive effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Immunosuppressive effects | ||||||||
| Factor Description | Immunosuppression is when your immune system is not functioning as it should. The immune system is made up of cells, tissues and organs that help the body fight off infections. If the immune system is suppressed, an infection that your body was able to control may become serious or even fatal. | ||||||||
| Mechanism Description |
|
||||||||
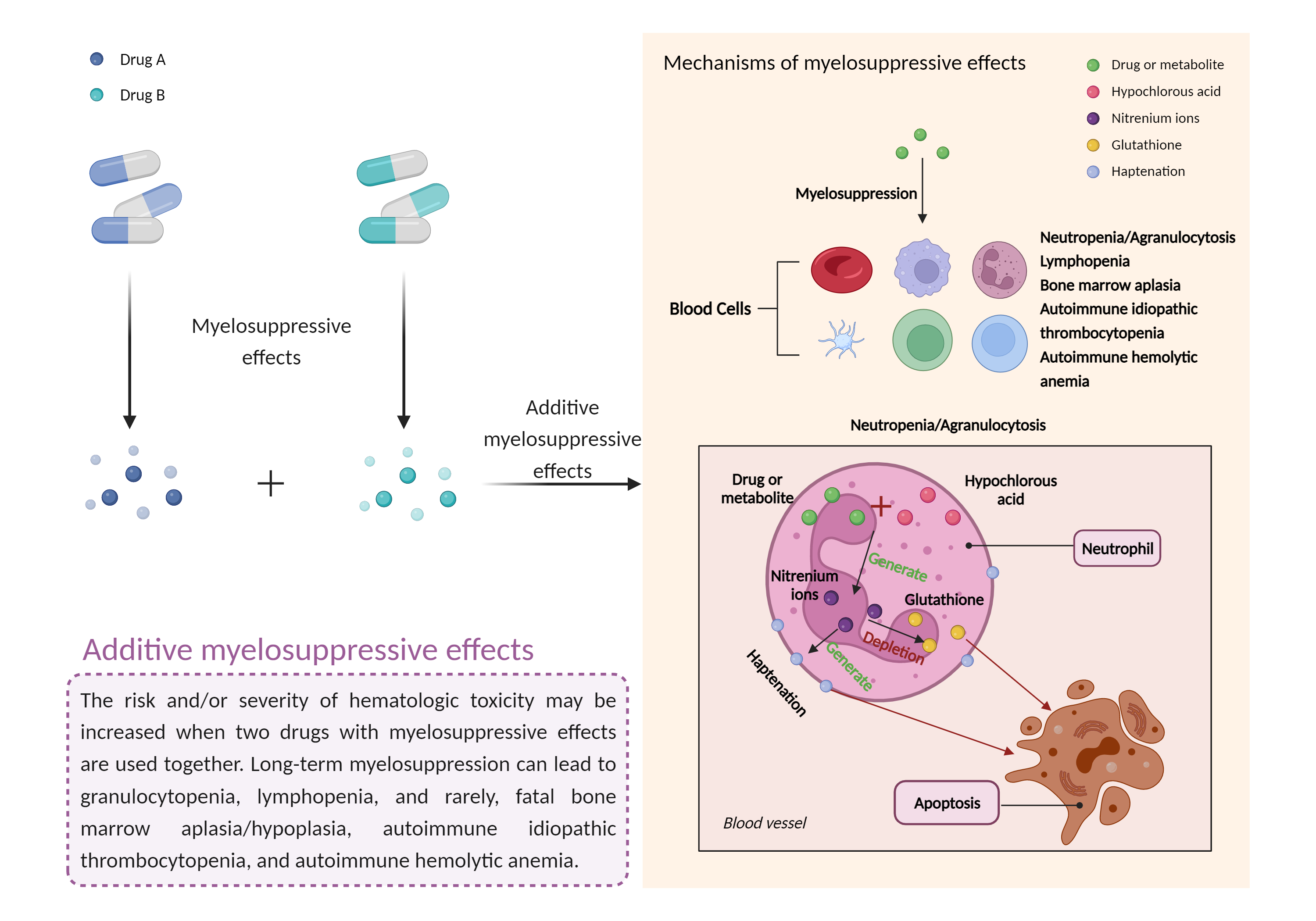
| Additive myelosuppressive effects Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Teriflunomide | Olaparib | |||||||
| Mechanism 2 | Myelosuppressive effects | Myelosuppressive effects | |||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Myelosuppressive effects | ||||||||
| Factor Description | Myelosuppression, also known as bone marrow suppression, is a decrease in bone marrow activity that leads to a decrease in the production of blood cells. Some blood cell disorders include: erythrocytopenia (anemia), leukopenia (neutropenia), and thrombocytopenia (thrombocytopenia). | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Close monitoring for the development of infection is recommended if leflunomide or teriflunomide is used in patients who are currently receiving or have recently received other immuno- or myelosuppressive agents, and vice versa. Platelet, white blood cell count, and hemoglobin or hematocrit should be evaluated at baseline and regularly during therapy. Patients should be advised to contact their physician if they develop signs and symptoms of infection such as fever, chills, diarrhea, sore throat, muscle aches, shortness of breath, blood in phlegm, weight loss, red or inflamed skin, body sores, and pain or burning during urination. If evidence of serious infection or bone marrow suppression occurs, treatment should be stopped, and cholestyramine or charcoal administered to accelerate elimination of leflunomide's active metabolite from plasma, which otherwise may take up to two years. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Arava (leflunomide). Hoechst Marion-Roussel Inc, Kansas City, MO. | ||||||||||||||||||
| 2 | Product Information. Aubagio (teriflunomide). Genzyme Corporation, Cambridge, MA. | ||||||||||||||||||
