| References |
| 1 |
Alffenaar JW, van Assen S, van der Werf TS, Kosterink JG, Uges DR "Omeprazole significantly reduces posaconazole serum trough level." Clin Infect Dis 48 (2009): 839. [PMID: 19220151]
|
| 2 |
Dolton MJ, Ray JE, Chen SC, Ng K, Pont L, McLachlan AJ "Multicenter study of posaconazole therapeutic drug monitoring: exposure-response and factors affecting concentration." Antimicrob Agents Chemother 56 (2012): 5503-10. [PMID: 22890761]
|
| 3 |
Krishna G, Abutarif M, Xuan F, Martinho M, Angulo D, Cornely OA "Pharmacokinetics of oral posaconazole in neutropenic patients receiving chemotherapy for acute myelogenous leukemia or myelodysplastic syndrome." Pharmacotherapy 28 (2008): 1223-32. [PMID: 18823218]
|
| 4 |
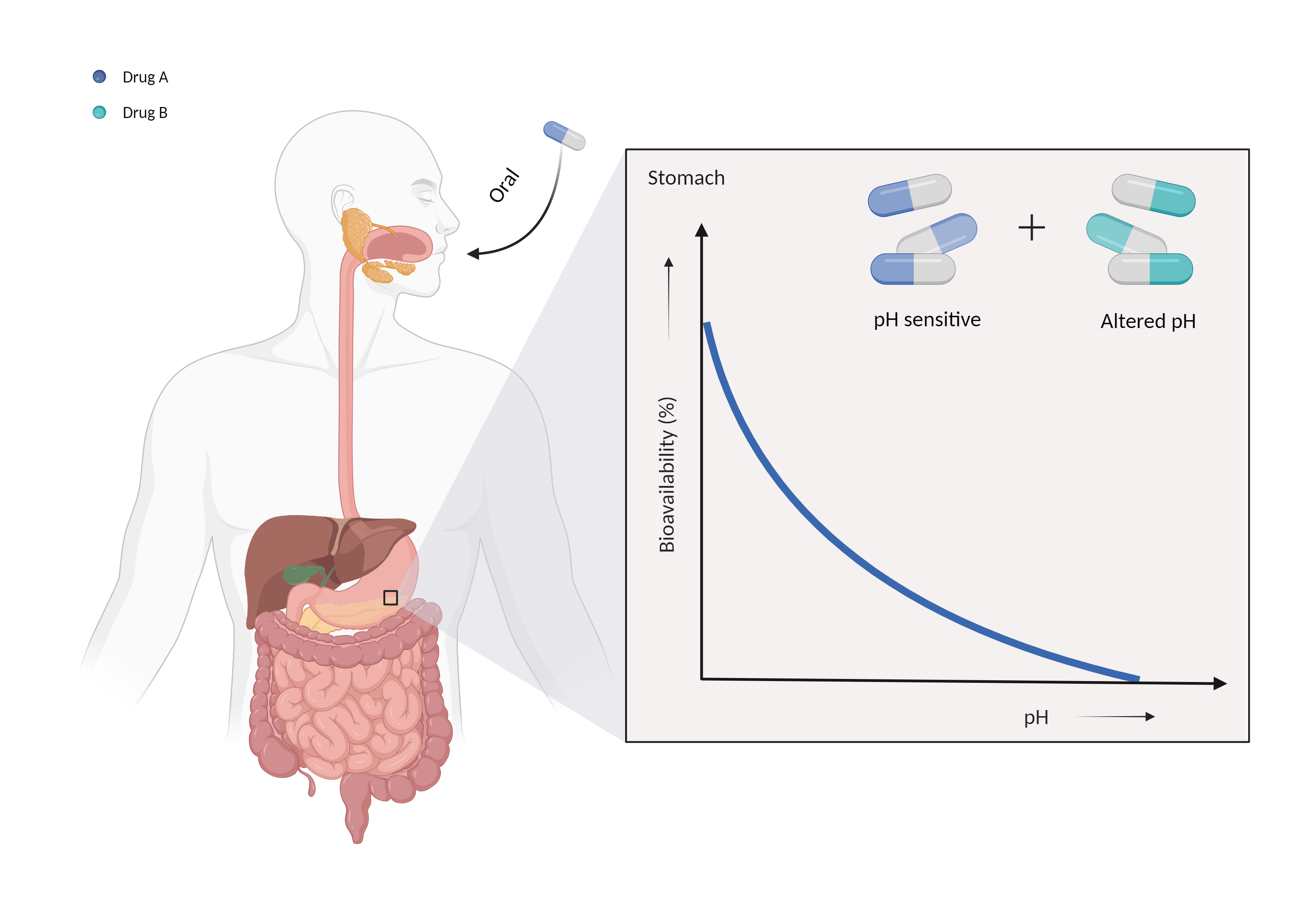
Krishna G, Moton A, Ma L, Medlock MM, McLeod J "The pharmacokinetics and absorption of posaconazole oral suspension under various gastric conditions in healthy volunteers." Antimicrob Agents Chemother 53 (2009): 958-66. [PMID: 19075045]
|
| 5 |
Neubauer WC, Engelhardt M, Konig A, et al. "Therapeutic drug monitoring of posaconazole in hematology patients: experience with a new high-performance liquid chromatography-based method." Antimicrob Agents Chemother 54 (2010): 4029-32. [PMID: 20547790]
|
| 6 |
Product Information. Noxafil (posaconazole). Schering-Plough Corporation, Kenilworth, NJ.
|
| 7 |
Shields RK, Clancy CJ, Vadnerkar A, et al "Posaconzaole serum concentrations among cardiothoracic transplant recipients: factors impacting trough levels and correlation with clinical response to therapy." Antimicrob Agents Chemother 55 (2011): 1308-11. [PMID: 21189337]
|
| 8 |
Vaes M, Hites M, Cotton F, et al. "Therapeutic drug monitoring of posaconazole in patients with acute myeloid leukemia or myelodysplasic syndrome." Antimicrob Agents Chemother 56 (2012): 6298-303. [PMID: 23027198]
|
| 9 |
Walravens J, Brouwers J, Spriet I, Tack J, Annaert P, Augustijns P "Effect of pH and Comedication on Gastrointestinal Absorption of Posaconazole: Monitoring of Intraluminal and Plasma Drug Concentrations." Clin Pharmacokinet 50 (2011): 725-34. [PMID: 21973269]
|
| 10 |
Krishna G, Moton A, Ma L, Malavade D, Medlock M, McLeod J "Effect of gastric pH, dosing regimen and prandial state, food and meal timing relative to dose, and gastro-intestinal motility on absorption and pharmacokinetics of the antifungal posaconazole." 18th European Congress of Clinical Microbiology and Infectious Diseases April (2008): 20.
|