Details of Drug-Drug Interaction
| Drug General Information (ID: DDI58DGYEU) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Trifluridine | Drug Info | Zidovudine | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antiviral Agents | Anti-Hiv Agents | |||||||
| Structure | |||||||||
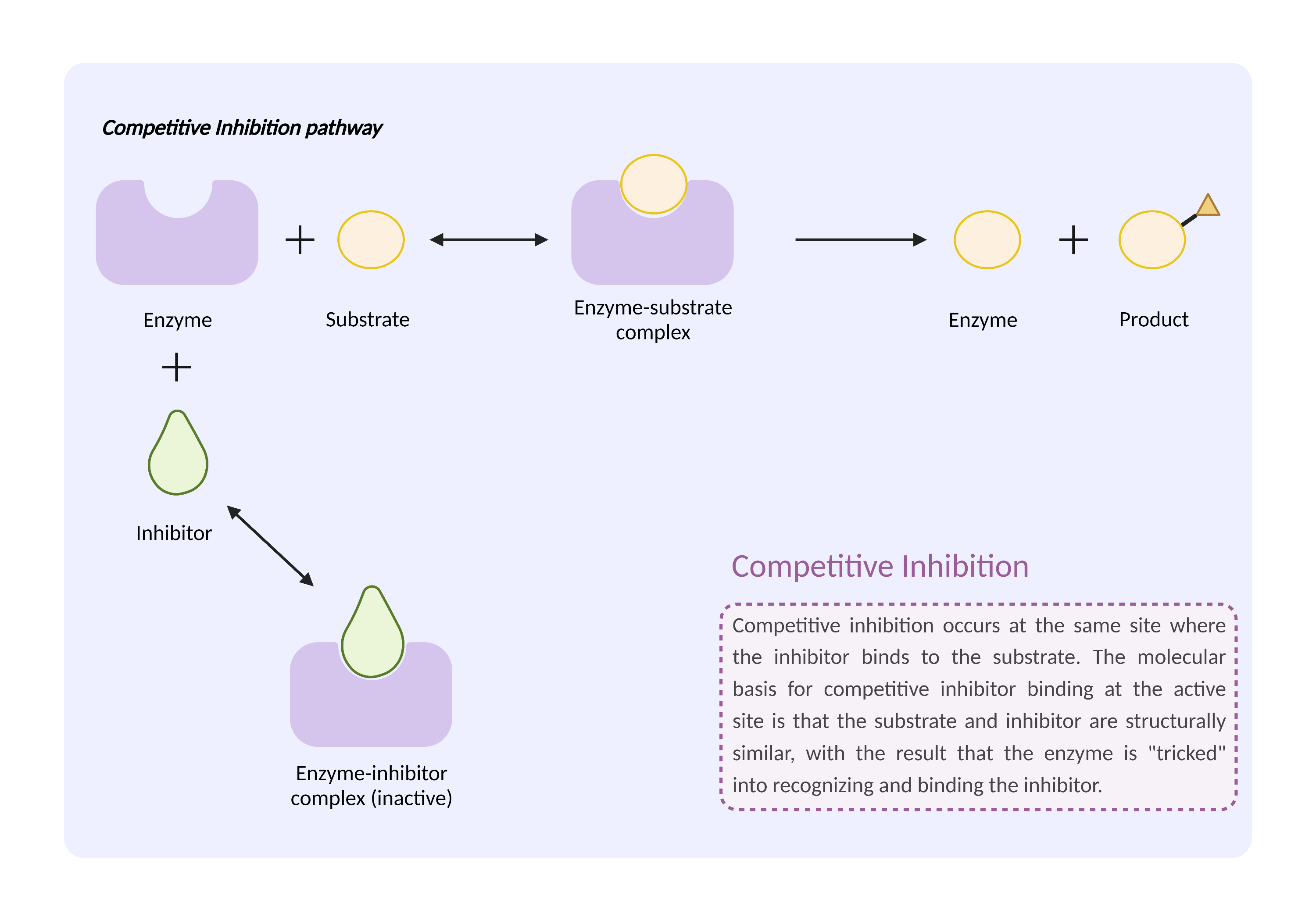
| Mechanism of Trifluridine-Zidovudine Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Competitive inhibition of metabolic enzyme Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Trifluridine | Zidovudine | |||||||
| Mechanism | Thymidine kinase substrate | Thymidine kinase substrate | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Thymidine kinase, cytosolic |
×
Structure
Sequence
MSCINLPTVLPGSPSKTRGQIQVILGPMFSGKSTELMRRVRRFQIAQYKCLVIKYAKDTRYSSSFCTHDRNTMEALPACLLRDVAQEALGVAVIGIDEGQFFPDIVEFCEAMANAGKTVIVAALDGTFQRKPFGAILNLVPLAESVVKLTAVCMECFREAAYTKRLGTEKEVEVIGGADKYHSVCRLCYFKKASGQPAGPDNKENCPVPGKPGEAVAARKLFAPQQILQCSPAN
|
|||||||
| Gene Name | TK1 | ||||||||
| Uniprot ID | KITH_HUMAN | ||||||||
| KEGG Pathway | hsa:7083 | ||||||||
| Protein Family | Thymidine kinase family | ||||||||
| Protein Function |
Cell-cycle-regulated enzyme of importance in nucleotide metabolism (PubMed:9575153). Catalyzes the first enzymatic step in the salvage pathway converting thymidine into thymidine monophosphate (PubMed:22385435). Transcriptional regulation limits expression to the S phase of the cell cycle and transient expression coincides with the oscillation in the intracellular dTTP concentration (Probable). Also important for the activation of anticancer and antiviral nucleoside analog prodrugs such as 1-b-d-arabinofuranosylcytosine (AraC) and 3c-azido-3c-deoxythymidine (AZT) (PubMed:22385435).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised if trifluridine is used concomitantly with drugs that are substrates of thymidine kinase. Patients should be monitored for loss of therapeutic efficacy. If the concomitant drug is an antiviral medication, patients should be monitored for loss of antiviral efficacy and an alternative antiviral agent that is not a thymidine kinase substrate, such as lamivudine, zalcitabine, didanosine and/or abacavir should be considered. | ||||||||
