Details of Drug-Drug Interaction
| Drug General Information (ID: DDI4G2IFZ8) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Topiramate | Drug Info | Valproic acid | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Anticonvulsants | Anticonvulsants | |||||||
| Structure | |||||||||
| Mechanism of Topiramate-Valproic acid Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
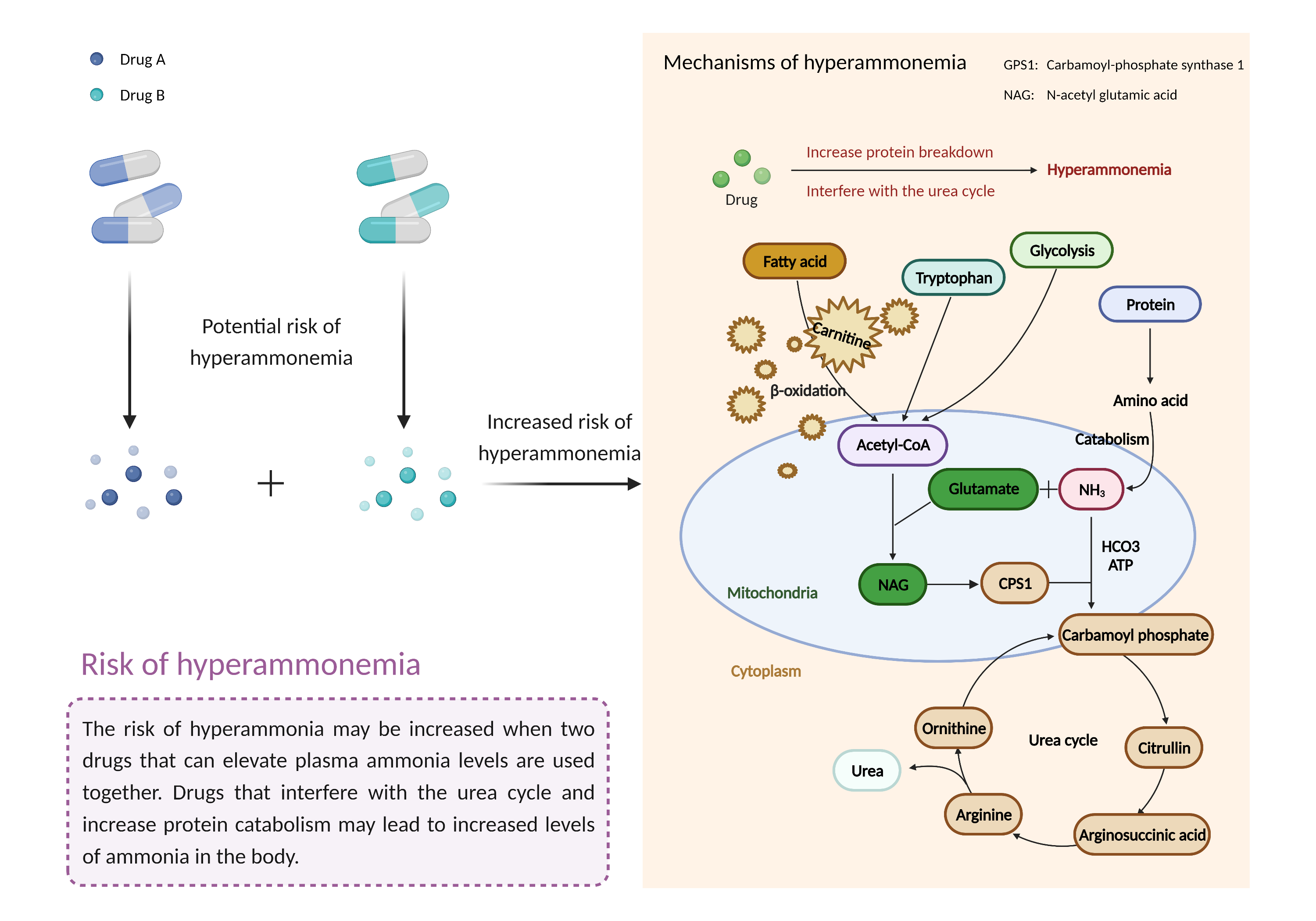
| Increased risk of hyperammonemia Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Topiramate | Valproic acid | |||||||
| Mechanism | Hyperammonemia | Hyperammonemia | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Hyperammonemia | ||||||||
| Factor Description | Hyperammonemia is a metabolic disorder characterized by elevated levels of ammonia. The most common manifestations of hyperammonemia are neurological signs and symptoms that may be acute or chronic, depending on the underlying abnormality, and severe life-threatening complications such as cerebral edema and brain herniation can occur. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Pharmacologic response to topiramate and valproic acid should be monitored more closely following addition or withdrawal of one or the other drug. Dose adjustments may be required if an interaction is suspected. Patients should be advised to notify their physician if they experience loss of seizure control. Patients should also be monitored for clinical symptoms of hyperammonemic encephalopathy, which often include acute alterations in level of consciousness and/or cognitive function with lethargy or vomiting. Monitoring of ammonia levels should be considered if such symptoms occur. Signs and symptoms usually abate with discontinuation of either drug. Discontinuation of topiramate or valproic acid should be considered if hypothermia occurs. Patients should be monitored for symptoms such as lethargy, confusion, coma, and cardiovascular or respiratory system changes. | ||||||||
