Details of Drug-Drug Interaction
| Drug General Information (ID: DDI4FBUXD3) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Zidovudine | Drug Info | Peginterferon beta-1a | Drug Info | |||||
| Drug Type | Small molecule | Interferons | |||||||
| Therapeutic Class | Anti-Hiv Agents | Interferons | |||||||
| Mechanism of Zidovudine-Peginterferon beta-1a Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
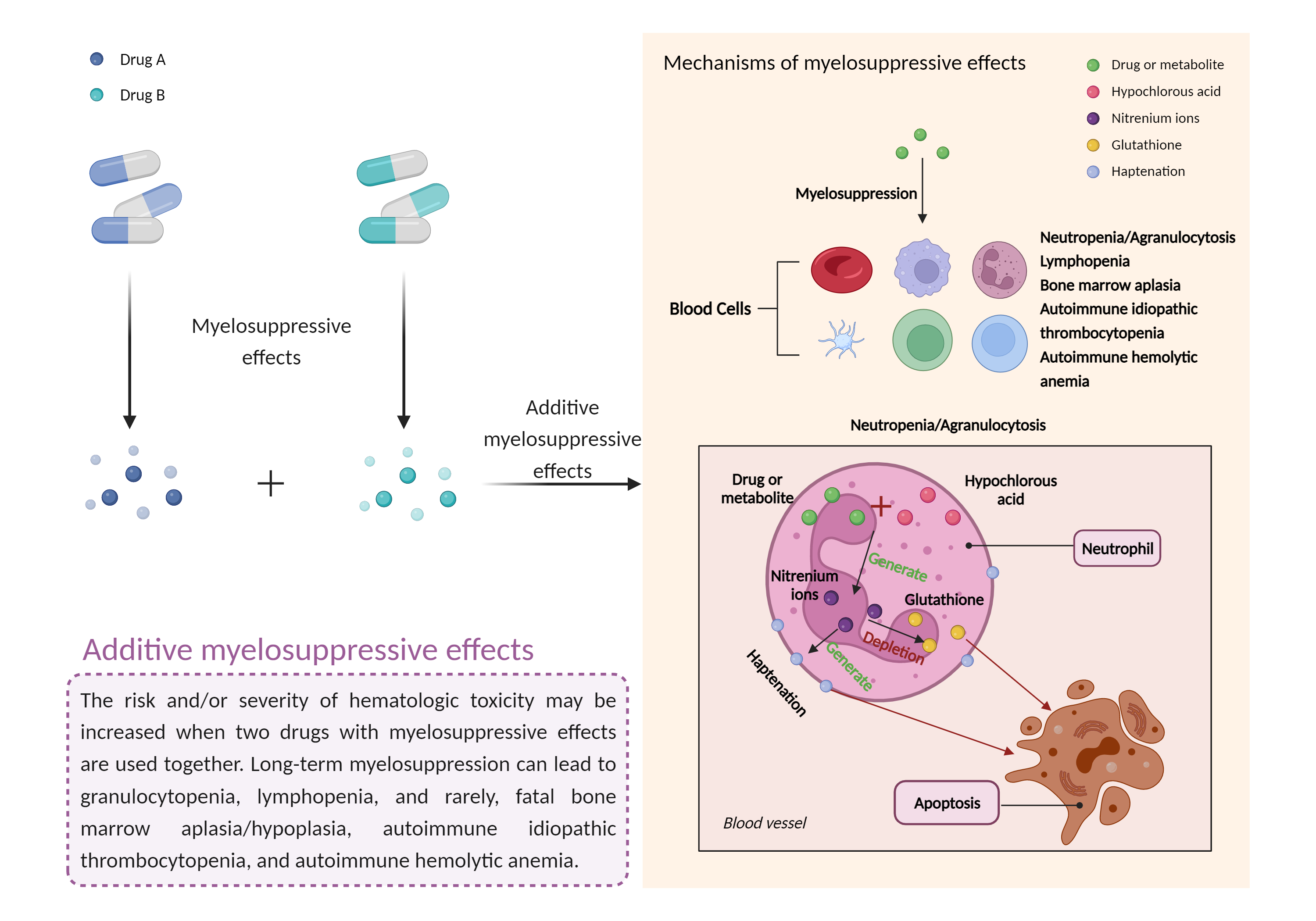
| Additive myelosuppressive effects Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Zidovudine | Peginterferon beta-1a | |||||||
| Mechanism | Myelosuppressive effects | Myelosuppressive effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Myelosuppressive effects | ||||||||
| Factor Description | Myelosuppression, also known as bone marrow suppression, is a decrease in bone marrow activity that leads to a decrease in the production of blood cells. Some blood cell disorders include: erythrocytopenia (anemia), leukopenia (neutropenia), and thrombocytopenia (thrombocytopenia). | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised if concomitant therapy is necessary, particularly with the alfa interferons. Complete blood counts, differential and platelet counts should be performed regularly and drug dosages adjusted accordingly. Patients should be advised to promptly report symptoms such as chills, fever, sore throat, pale skin, or unusual fatigue to their physician. Cessation of one or all drugs may be necessary if toxicity develops. | ||||||||
