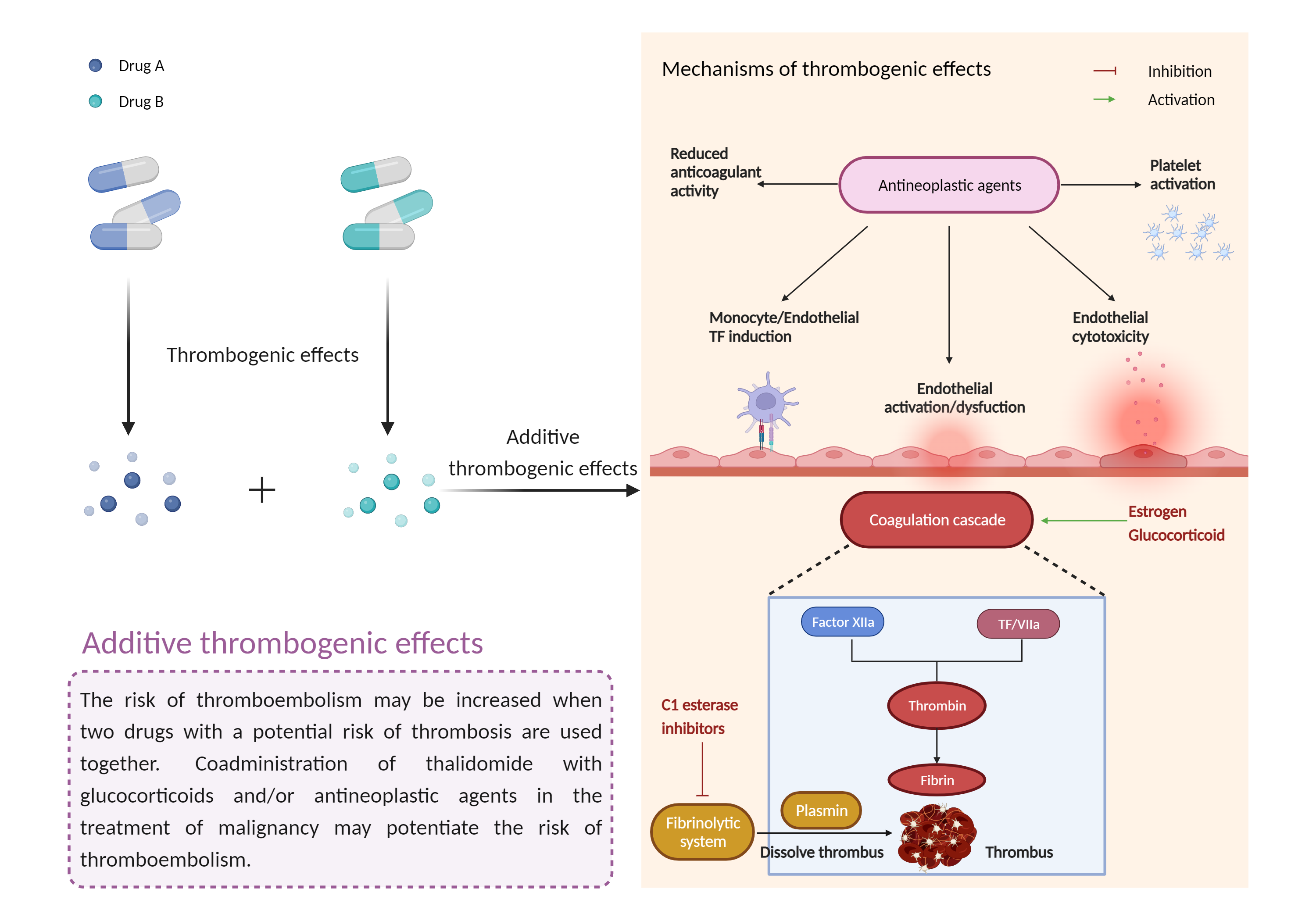
| Management |
The use of estrogen-containing medications including combined oral contraceptive pills should be undertaken with caution in multiple myeloma patients receiving thalidomide with standard chemotherapeutic agents and/or steroids. Patients should be apprised of potentially increased risk of venous thromboembolic events if a combined oral contraceptive pill is chosen as one of two effective methods of contraception that must be used simultaneously and continuously for 4 weeks before, during (even in case of dose interruption), and for 4 weeks after thalidomide therapy. Input from a gynecologist or similar expert on adequate contraception should be sought as needed. Patients should be advised to seek medical attention if they develop potential signs and symptoms of thromboembolism such as chest pain, shortness of breath, and pain or swelling in the arms or legs. Prophylaxis with anticoagulants such as low-molecular weight heparins or warfarin may be appropriate, but the decision to take thromboprophylactic measures should be made after careful assessment of underlying risk factors. If a thromboembolic event occurs during therapy with thalidomide, treatment must be discontinued and standard anticoagulation therapy started. Once anticoagulation is stabilized and complications of the thromboembolic event under control, thalidomide may be restarted at the original dose if benefit is deemed to outweigh the risks. Anticoagulation therapy should be continued during the remaining course of thalidomide treatment. Because some patients receiving thalidomide may also develop sudden, severe neutropenia and/or thrombocytopenia, hormonal contraceptives that are implanted may carry an increased risk for infection or bleeding either at insertion, removal, or during use. |