Details of Drug-Drug Interaction
| Drug General Information (ID: DDI3SPONLQ) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Physostigmine | Drug Info | Brigatinib | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Ophthalmic Glaucoma Agents/Antidotes | Multikinase Inhibitors | |||||||
| Structure | |||||||||
| Mechanism of Physostigmine-Brigatinib Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
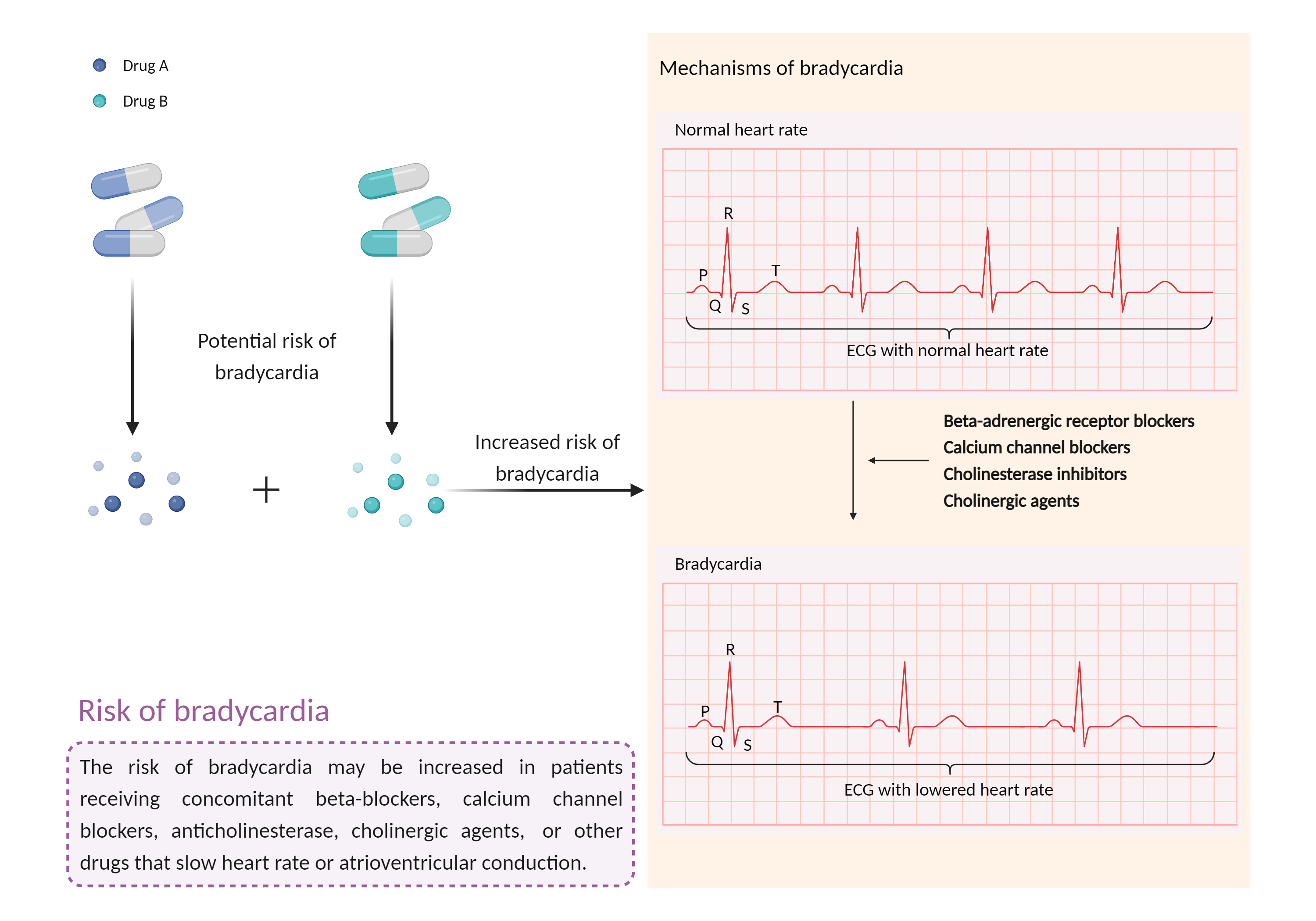
| Increased risk of bradycardia Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Physostigmine | Brigatinib | |||||||
| Mechanism | Bradycardia | Bradycardia | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Bradycardia | ||||||||
| Factor Description | Bradycardia is a slow heart rate in which the heart beats less than 60 times per minute. If the heart rate is very slow and the heart is not pumping enough oxygen-rich blood to the body, and you may feel dizzy, very tired or weak, and short of breath. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised if brigatinib is used concomitantly with beta-blockers, calcium channel blockers, digitalis, or other drugs that can slow the heart rate or atrioventricular conduction such as alectinib, atazanavir, fingolimod, flecainide, ivabradine, lacosamide, lithium, mefloquine, moricizine, propafenone, succinylcholine, thalidomide, H2-receptor antagonists, tricyclic antidepressants, and anticholinesterase or cholinergic agents. Heart rate and blood pressure should be monitored frequently, and patients should be advised to seek medical attention if they experience dizziness, lightheadedness, fainting, or irregular heartbeat. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Alunbrig (brigatinib). Ariad Pharmaceuticals Inc, Cambridge, MA. | ||||||||||||||||||
