Details of Drug-Drug Interaction
| Drug General Information (ID: DDI3PDKMUF) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Siponimod | Drug Info | Mogamulizumab | Drug Info | |||||
| Drug Type | Small molecule | Monoclonal antibody | |||||||
| Therapeutic Class | Selective Immunosuppressants | Antineoplastics | |||||||
| Mechanism of Siponimod-Mogamulizumab Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Additive immunosuppressive effects Click to Show/Hide Mechanism Graph | |||||||||
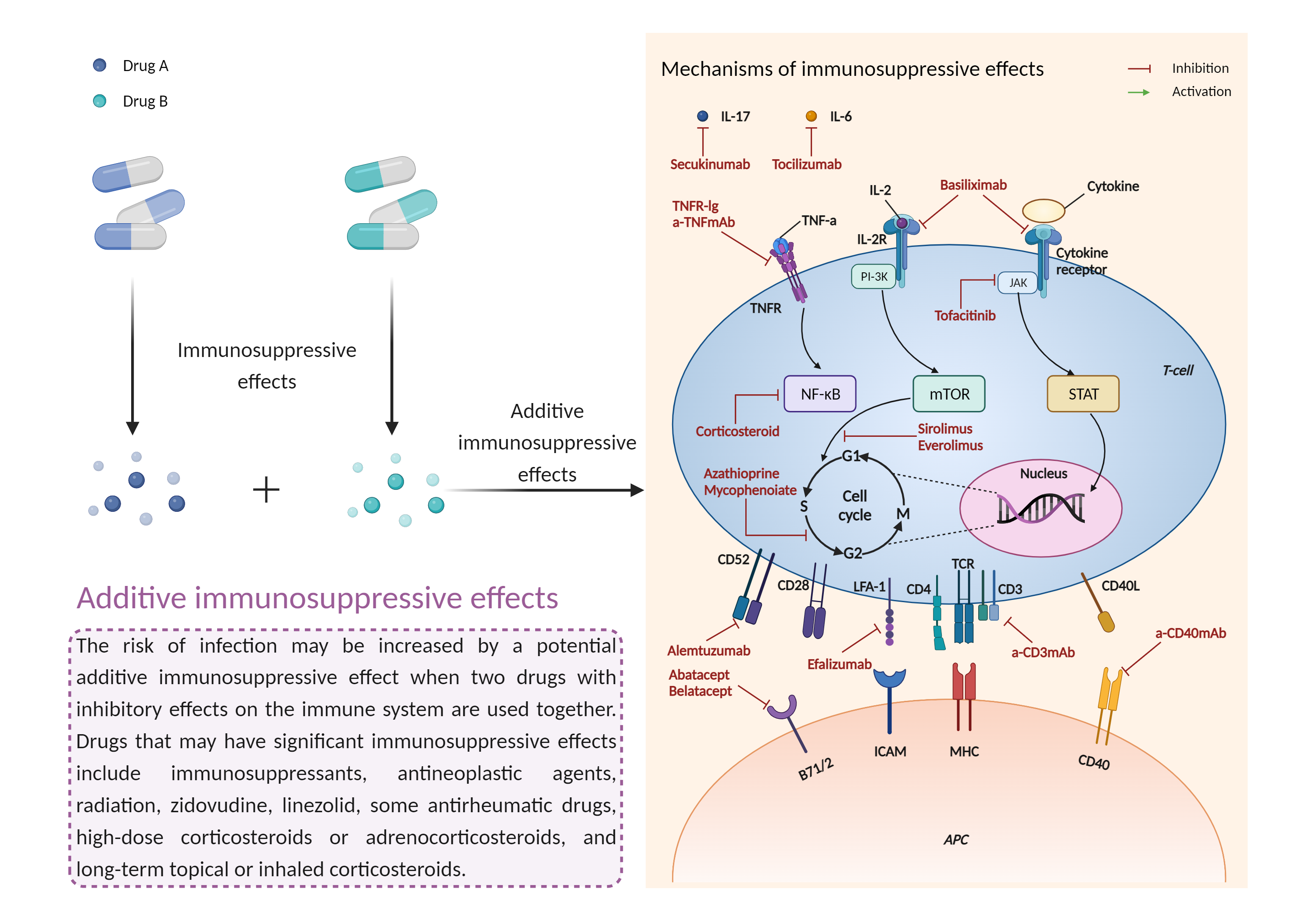 |
|||||||||
| Drug Name | Siponimod | Mogamulizumab | |||||||
| Mechanism | Immunosuppressive effects | Immunosuppressive effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Immunosuppressive effects | ||||||||
| Factor Description | Immunosuppression is when your immune system is not functioning as it should. The immune system is made up of cells, tissues and organs that help the body fight off infections. If the immune system is suppressed, an infection that your body was able to control may become serious or even fatal. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | The safety and efficacy of siponimod in combination with antineoplastic, immunosuppressive, or immune-modulating agents have not been evaluated. Caution is advised during coadministration and for 3 to 4 weeks after the last dose of siponimod. When switching from drugs with prolonged immune effects to siponimod, the half-life and mode of action of these drugs must be considered to avoid unintended additive immunosuppressive effects while at the same time minimizing risk of disease reactivation. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Cerner Multum, Inc. "Australian Product Information.". | ||||||||||||||||||
| 2 | Product Information. Mayzent (siponimod). Novartis Pharmaceuticals, East Hanover, NJ. | ||||||||||||||||||
