| Management |
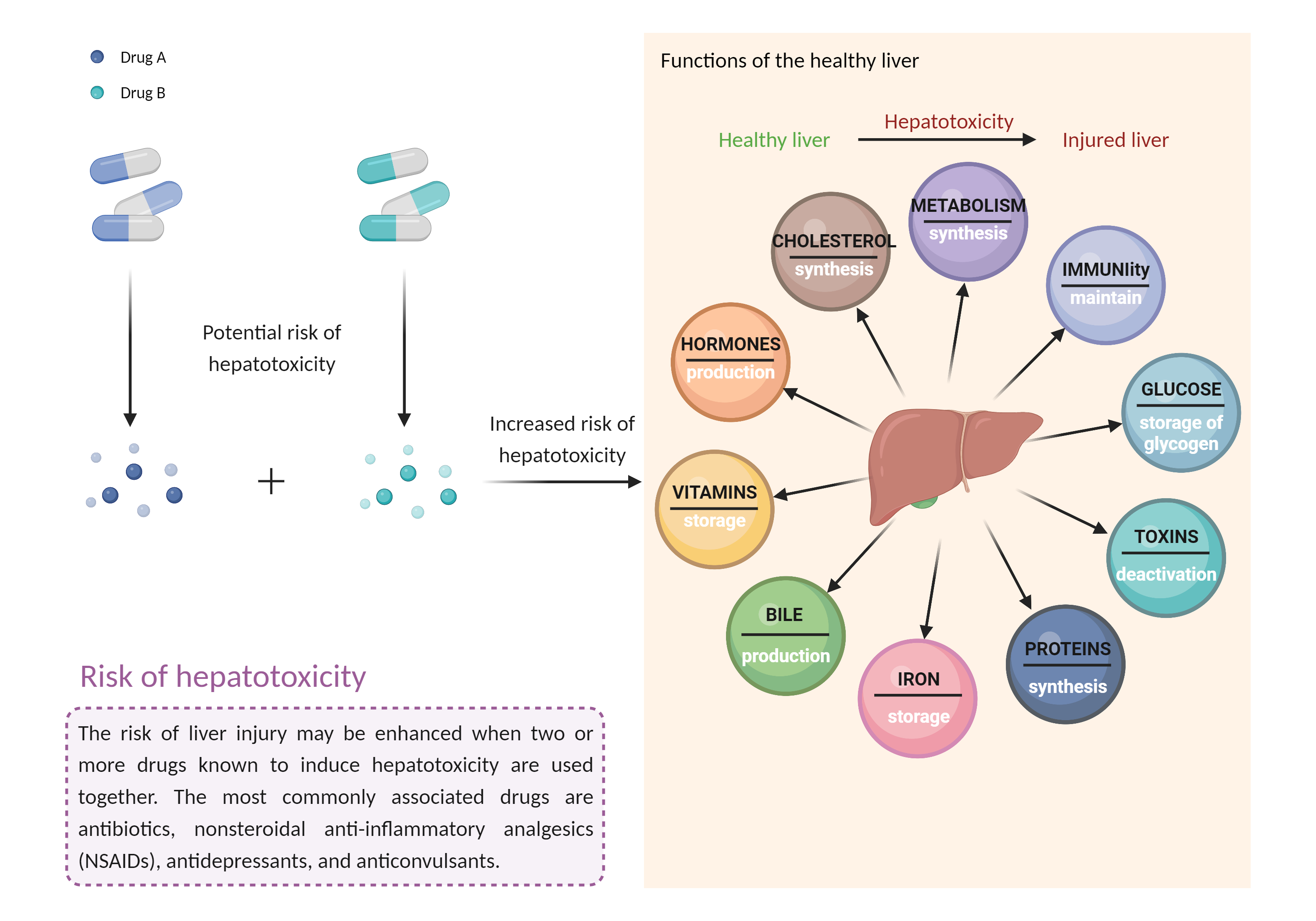
Caution is advised if leflunomide or teriflunomide must be used in patients who are currently receiving or have recently received treatment with other hepatotoxic agents (e.g., acetaminophen alcohol androgens and anabolic steroids antituberculous agents azole antifungal agents ACE inhibitors cyclosporine (high dosages) disulfiram endothelin receptor antagonists interferons ketolide and macrolide antibiotics kinase inhibitors minocycline nonsteroidal anti-inflammatory agents nucleoside reverse transcriptase inhibitors proteasome inhibitors retinoids thiazolidinediones tolvaptan vincristine zileuton anticonvulsants such as carbamazepine, hydantoins, felbamate, and valproic acid lipid-lowering medications such as fenofibrate, lomitapide, mipomersen, niacin, and statins herbals and nutritional supplements such as black cohosh, chaparral, comfrey, DHEA, kava, pennyroyal oil, and red yeast rice), and vice versa. Liver enzymes and bilirubin should be measured prior to initiation of leflunomide/teriflunomide therapy and at least monthly for the first six months of treatment and every 6 to 8 weeks thereafter. Patients with preexisting liver disease or elevated baseline liver enzymes (i.e., ALT greater than two times ULN) should not receive leflunomide or teriflunomide. Patients who develop elevated serum ALT greater than three times ULN while receiving these medications should discontinue treatment and be given washout procedures with cholestyramine or activated charcoal to accelerate elimination of leflunomide's active metabolite from plasma, which otherwise may take up to two years. Follow-up monitoring should be conducted at least weekly until the ALT value is within normal range, and washout procedures repeated as necessary. All patients treated with leflunomide or teriflunomide should be advised to seek medical attention if they experience potential signs and symptoms of hepatotoxicity such as fever, rash, itching, anorexia, nausea, vomiting, fatigue, malaise, right upper quadrant pain, dark urine, pale stools, and jaundice. |