Details of Drug-Drug Interaction
| Drug General Information (ID: DDI3I8AWXP) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Carmustine | Drug Info | Cimetidine | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Alkylating Agents | Antiulcer Agents | |||||||
| Structure | |||||||||
| Mechanism of Carmustine-Cimetidine Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
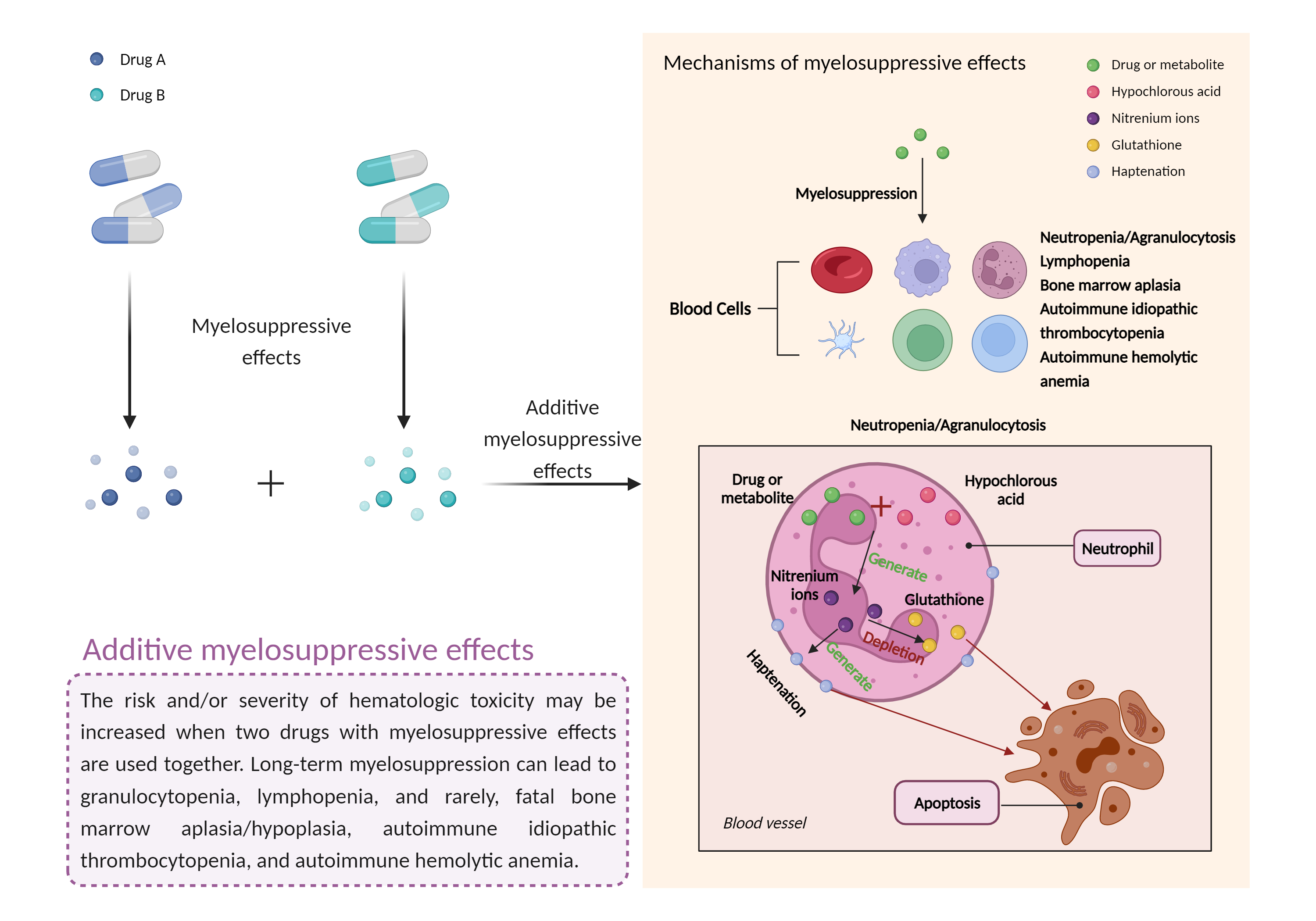
| Additive myelosuppressive effects Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Carmustine | Cimetidine | |||||||
| Mechanism | Myelosuppressive effects | Myelosuppressive effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Myelosuppressive effects | ||||||||
| Factor Description | Myelosuppression, also known as bone marrow suppression, is a decrease in bone marrow activity that leads to a decrease in the production of blood cells. Some blood cell disorders include: erythrocytopenia (anemia), leukopenia (neutropenia), and thrombocytopenia (thrombocytopenia). | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised if systemic carmustine is used in combination with cimetidine. Patients should be closely monitored for the development of delayed myelosuppression that may occur 4 to 6 weeks after carmustine administration. Blood counts are recommended weekly for at least 6 weeks after a dose. alternatively, other H2-receptor antagonists such as ranitidine and famotidine may be considered in patients who require treatment with carmustine, since they have not been reported to cause the interaction and generally have minimal effects on hepatic metabolism. | ||||||||
