Details of Drug-Drug Interaction
| Drug General Information (ID: DDI30MJUSB) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Loperamide | Drug Info | Saquinavir | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antidiarrheals | Anti-Hiv Agents | |||||||
| Structure | |||||||||
| Mechanism of Loperamide-Saquinavir Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
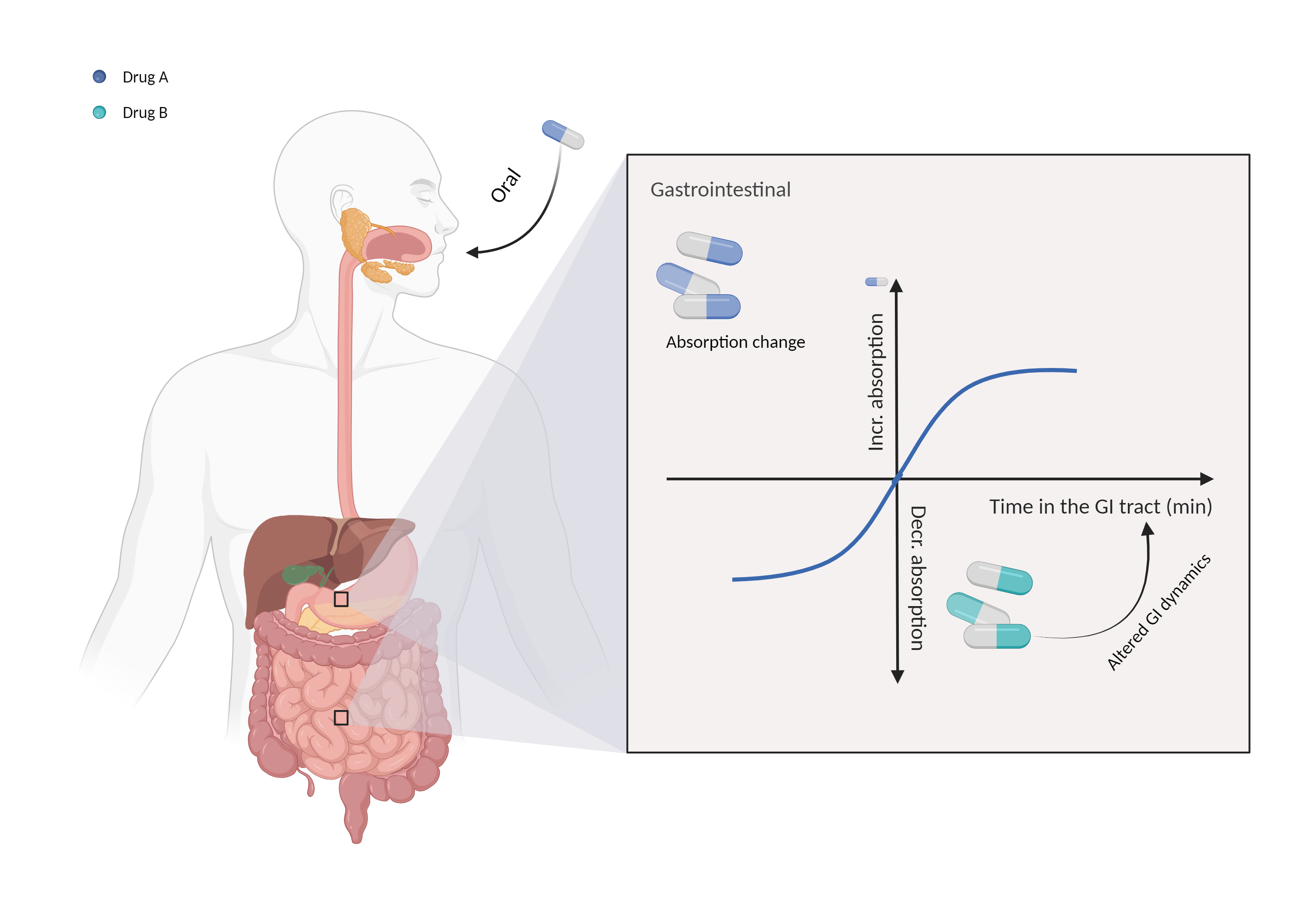
| Altered gastrointestinal dynamics Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Loperamide | Saquinavir | |||||||
| Mechanism 1 | Decrease gastrointestinal motility | Absorption is influenced by gastrointestinal motility | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Gastrointestinal Motility | ||||||||
| Factor Description | Gastrointestinal motility is an important factor in determining the absorption of orally administered drugs, and it controls the residence time of the drug in the digestive tract. | ||||||||
| Mechanism Description |
|
||||||||
| Increased risk of prolong QT interval Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Loperamide | Saquinavir | |||||||
| Mechanism 2 | Prolong QT interval | Prolong QT interval | |||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | QT interval | ||||||||
| Factor Description | Long QT syndrome is a heart signaling disorder that can cause a fast, chaotic heartbeat (arrhythmia). Many people may not exhibit symptoms, and usually the condition is detected during routine medical tests. In others, the most common symptoms include: sudden fainting, palpitations, dizziness, seizures, sudden death. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Given the risk of reduced viral susceptibility and resistance development associated with subtherapeutic antiretroviral drug levels, prolonged use of loperamide should preferably be avoided in patients treated with saquinavir. However, the effect of loperamide on the pharmacokinetics of ritonavir-boosted saquinavir is unknown. Caution is recommended if coadministration is required. Patients should be counseled to not exceed the recommended dosage and frequency or duration of use of loperamide, and to seek prompt medical attention if they experience symptoms that could indicate the occurrence of torsade de pointes such as dizziness, lightheadedness, fainting, palpitation, irregular heart rhythm, shortness of breath, or syncope. If loperamide-induced cardiotoxicity is suspected, promptly discontinue loperamide and initiate therapy to manage and prevent cardiac arrhythmias and adverse outcomes. Electrical pacing or cardioversion may be necessary if torsade de pointes persists despite pharmacotherapy. In many of the reported cases of loperamide-induced cardiotoxicity, standard antiarrhythmic drugs were ineffective, and electrical pacing or cardioversion was necessary. | ||||||||
