Details of Drug-Drug Interaction
| Drug General Information (ID: DDI298RD3L) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Nimodipine | Drug Info | Nesiritide | Drug Info | |||||
| Drug Type | Small molecule | Hormones | |||||||
| Therapeutic Class | Vasodilator Agents | Recombinant Natriuretic Peptide | |||||||
| Structure | |||||||||
| Mechanism of Nimodipine-Nesiritide Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Additive hypotensive effects Click to Show/Hide Mechanism Graph | |||||||||
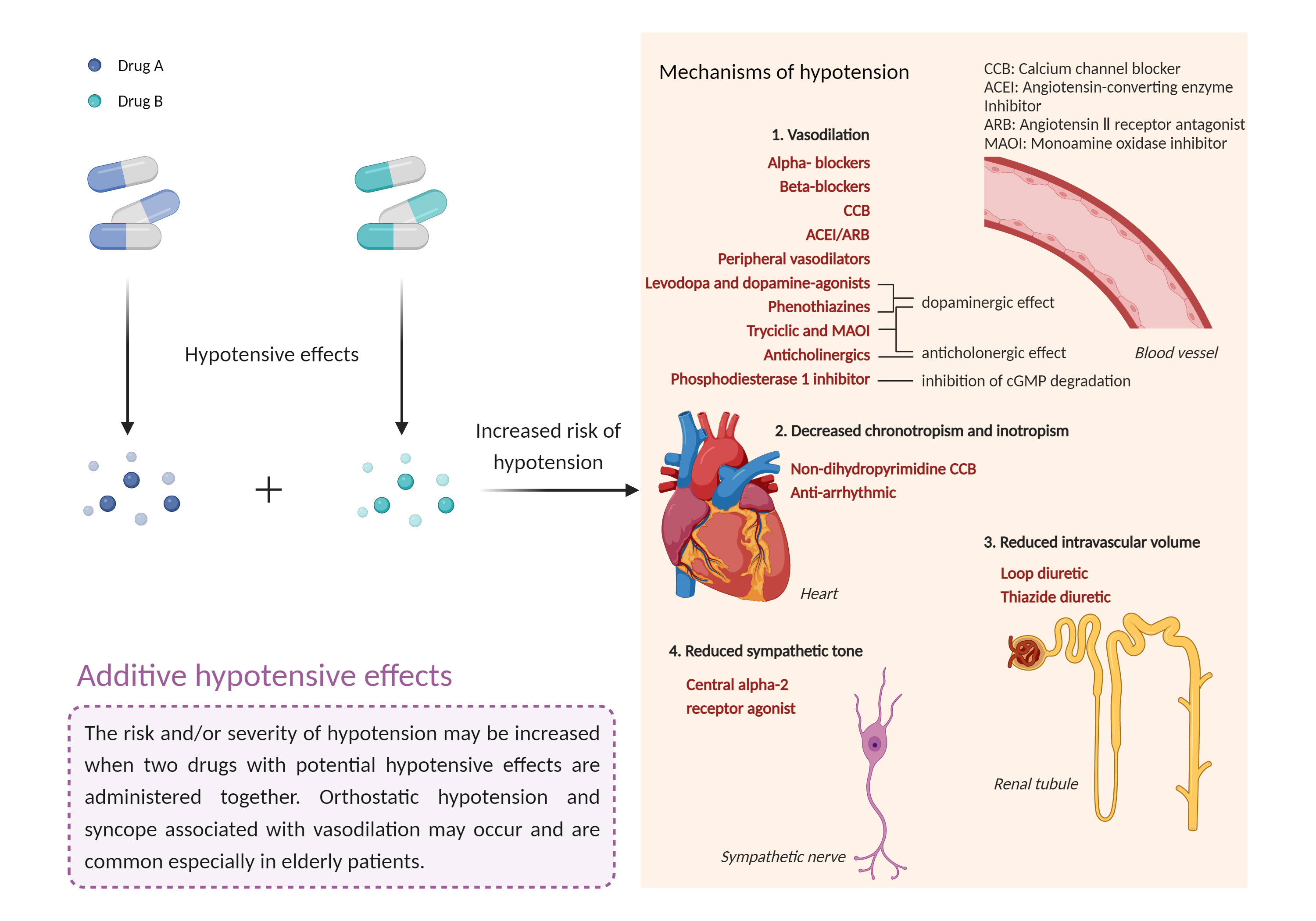 |
|||||||||
| Drug Name | Nimodipine | Nesiritide | |||||||
| Mechanism |
Antihypertensive agent Calcium channel unspecific Blocker |
Antihypertensive agent Guanylate cyclase Agonist |
|||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Voltage-dependent L-type calcium channel | Structure Sequence | |||||||
| Protein Family | Calcium channel beta subunit family | ||||||||
| Protein Function |
Regulatory subunit of L-type calcium channels (PubMed:1309651, PubMed:8107964, PubMed:15615847). Regulates the activity of L-type calcium channels that contain CACNA1A as pore-forming subunit (By similarity). Regulates the activity of L-type calcium channels that contain CACNA1C as pore-forming subunit and increases the presence of the channel complex at the cell membrane (PubMed:15615847). Required for functional expression L-type calcium channels that contain CACNA1D as pore-forming subunit (PubMed:1309651). Regulates the activity of L-type calcium channels that contain CACNA1B as pore-forming subunit (PubMed:8107964).
Click to Show/Hide
|
||||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Guanylate cyclase soluble | Structure Sequence | |||||||
| Protein Family | Adenylyl cyclase class-4/guanylyl cyclase family | ||||||||
| Protein Function |
There are two types of guanylate cyclases: soluble forms and membrane-associated receptor forms. Activated by nitric oxide in the presence of magnesium or manganese ions.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Whether used alone or with other agents, nesiritide should be administered only in settings where blood pressure can be closely monitored. The dosage should be reduced or the drug discontinued in patients who develop hypotension. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Natrecore (nesiritide) Scios Inc, Sunnyvale, CA. | ||||||||||||||||||
