Details of Drug-Drug Interaction
| Drug General Information (ID: DDI28TZR3J) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Nitroprusside | Drug Info | Maraviroc | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antihypertensive Agents | Anti-Hiv Agents | |||||||
| Structure | |||||||||
| Mechanism of Nitroprusside-Maraviroc Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Additive hypotensive effects Click to Show/Hide Mechanism Graph | |||||||||
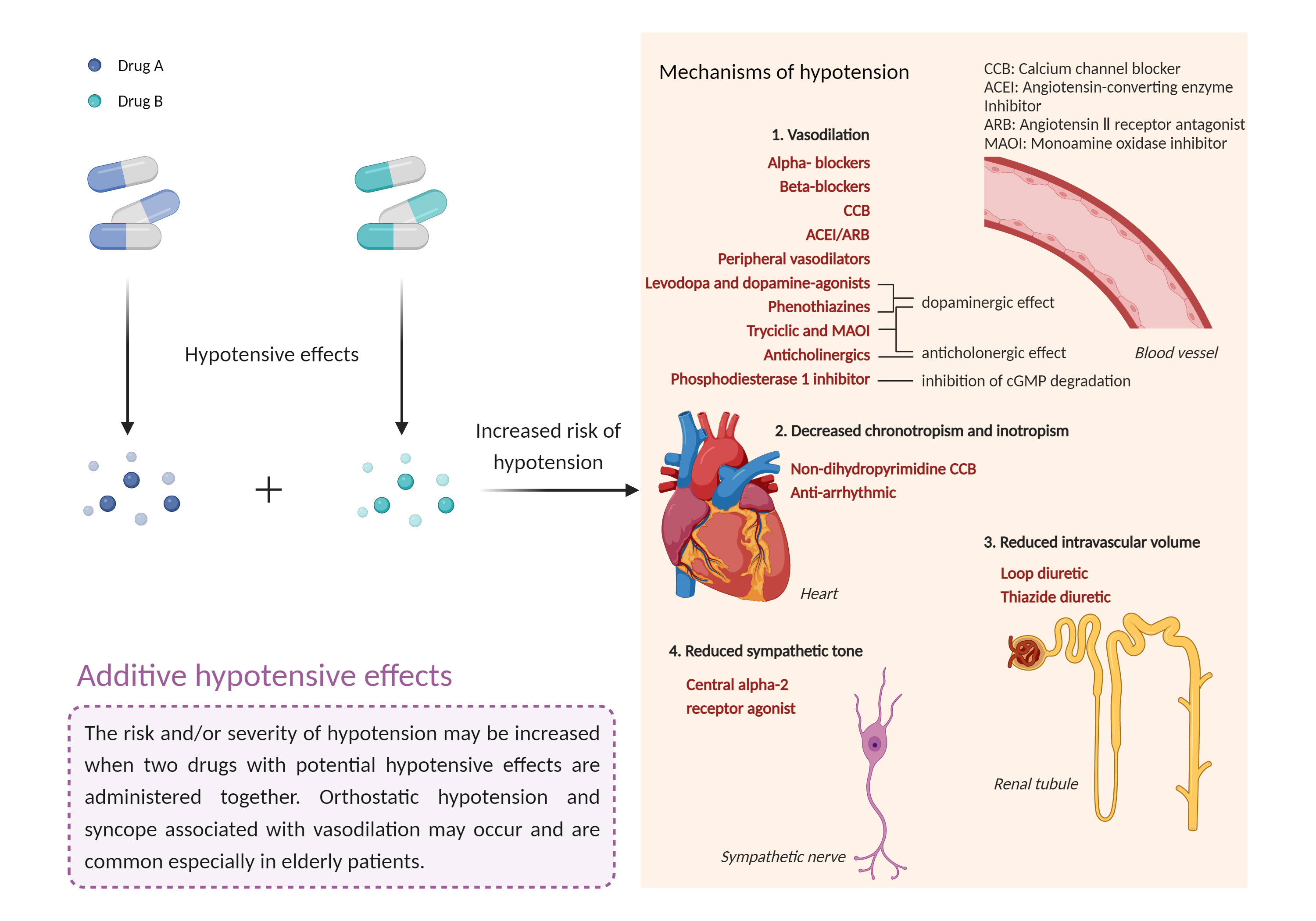 |
|||||||||
| Drug Name | Nitroprusside | Maraviroc | |||||||
| Mechanism 1 |
Antihypertensive agent Guanylate cyclase Agonist |
Antihypertensive agent | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Guanylate cyclase soluble | Structure Sequence | |||||||
| Protein Family | Adenylyl cyclase class-4/guanylyl cyclase family | ||||||||
| Protein Function |
There are two types of guanylate cyclases: soluble forms and membrane-associated receptor forms. Activated by nitric oxide in the presence of magnesium or manganese ions.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Mechanism 2 |
Antihypertensive agent Guanylate cyclase Agonist |
Hypotensive effects | |||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Guanylate cyclase soluble | Structure Sequence | |||||||
| Protein Family | Adenylyl cyclase class-4/guanylyl cyclase family | ||||||||
| Protein Function |
There are two types of guanylate cyclases: soluble forms and membrane-associated receptor forms. Activated by nitric oxide in the presence of magnesium or manganese ions.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised when maraviroc is prescribed to patients on concomitant medications known to lower blood pressure. Close monitoring for development of hypotension is recommended. Ambulatory patients should be advised to avoid rising abruptly from a sitting or recumbent position and to notify their doctor if they experience dizziness, lightheadedness, syncope, orthostasis, or tachycardia. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Selzentry (maraviroc). Pfizer U.S. Pharmaceuticals Group, New York, NY. | ||||||||||||||||||
