Details of Drug-Drug Interaction
| Drug General Information (ID: DDI26IGTRX) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Phenytoin | Drug Info | Sulfinpyrazone | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Anticonvulsants | Uricosuric Agents | |||||||
| Structure | |||||||||
| Mechanism of Phenytoin-Sulfinpyrazone Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
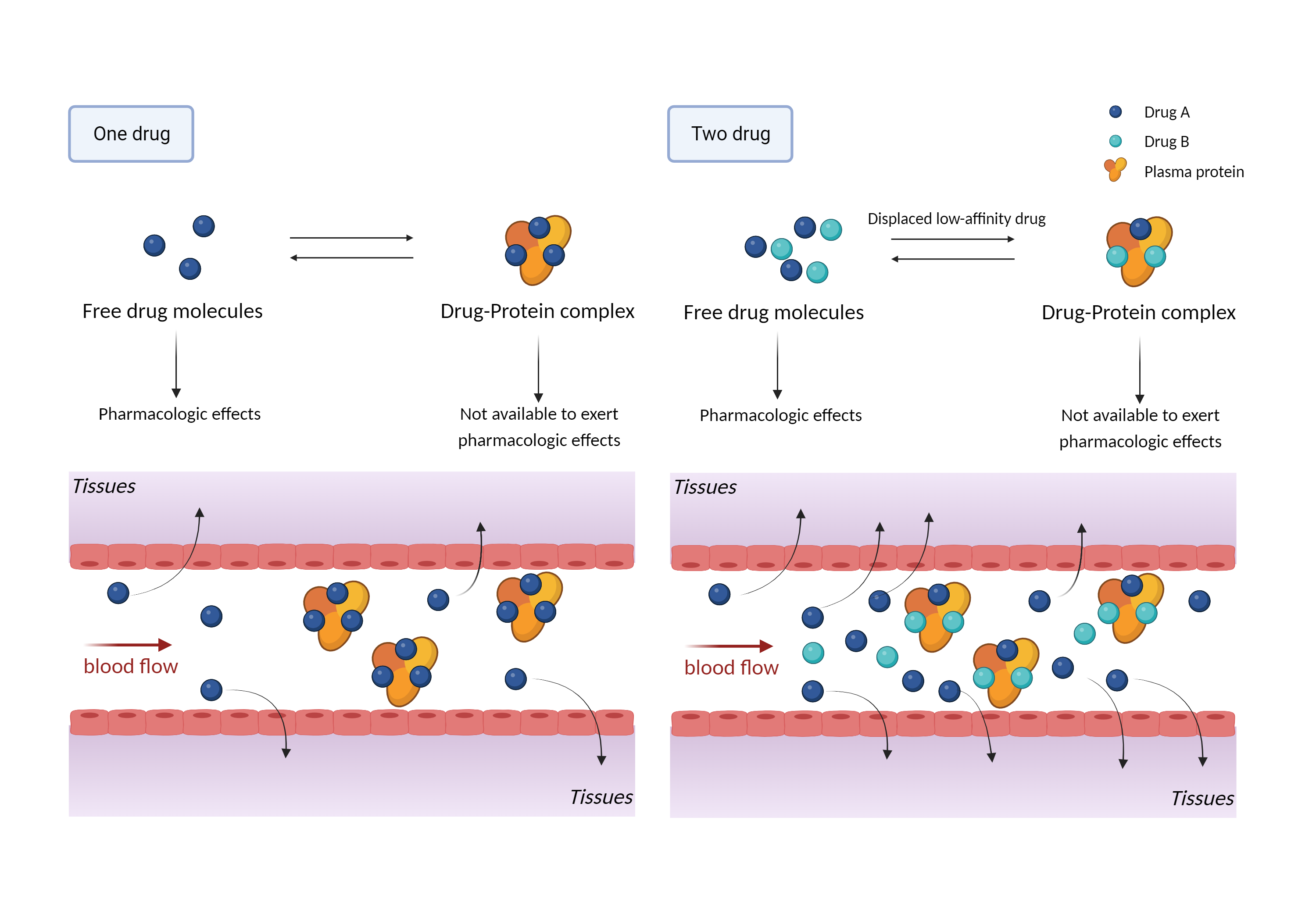
| Competitive binding of plasma proteins Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Phenytoin | Sulfinpyrazone | |||||||
| Mechanism 1 | Competition for plasma protein binding sites | Competition for plasma protein binding sites | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Plasma protein binding | ||||||||
| Factor Description | Plasma protein binding refers to the degree of binding of a drug to proteins in the blood, and the lower the degree of binding, the more efficiently the drug can cross cell membranes or diffuse. Plasma protein binding alters the distribution and therapeutic characteristics of the drugs. | ||||||||
| Mechanism Description |
|
||||||||
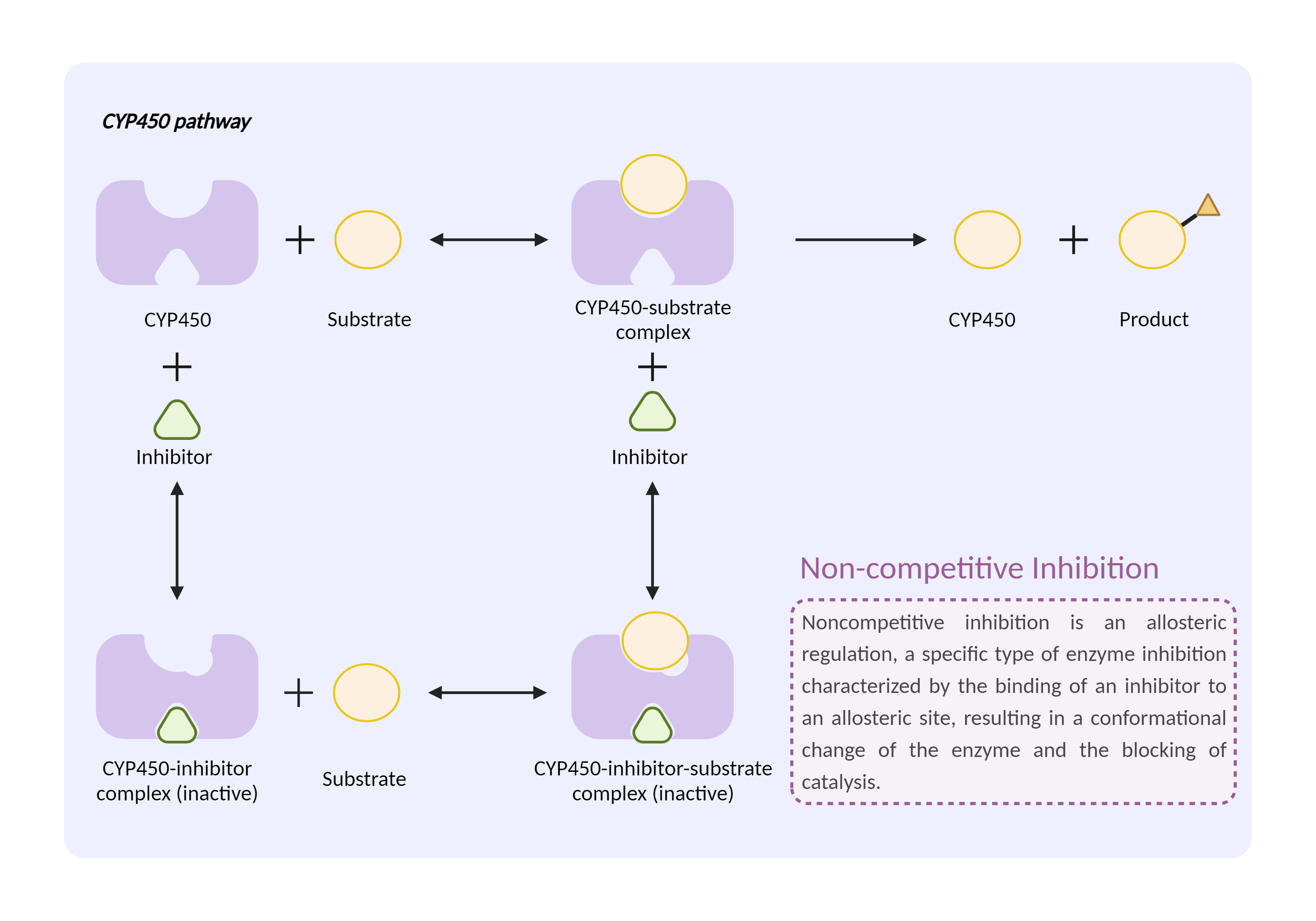
| CYP450 enzyme inhibition Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Phenytoin | Sulfinpyrazone | |||||||
| Mechanism 2 | CYP450 2C9 substrate | CYP450 2C9 inhibitor | |||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Cytochrome P450 2C9 |
×
Structure
Sequence
MDSLVVLVLCLSCLLLLSLWRQSSGRGKLPPGPTPLPVIGNILQIGIKDISKSLTNLSKVYGPVFTLYFGLKPIVVLHGYEAVKEALIDLGEEFSGRGIFPLAERANRGFGIVFSNGKKWKEIRRFSLMTLRNFGMGKRSIEDRVQEEARCLVEELRKTKASPCDPTFILGCAPCNVICSIIFHKRFDYKDQQFLNLMEKLNENIKILSSPWIQICNNFSPIIDYFPGTHNKLLKNVAFMKSYILEKVKEHQESMDMNNPQDFIDCFLMKMEKEKHNQPSEFTIESLENTAVDLFGAGTETTSTTLRYALLLLLKHPEVTAKVQEEIERVIGRNRSPCMQDRSHMPYTDAVVHEVQRYIDLLPTSLPHAVTCDIKFRNYLIPKGTTILISLTSVLHDNKEFPNPEMFDPHHFLDEGGNFKKSKYFMPFSAGKRICVGEALAGMELFLFLTSILQNFNLKSLVDPKNLDTTPVVNGFASVPPFYQLCFIPV
|
|||||||
| Gene Name | CYP2C9 | ||||||||
| Uniprot ID | CP2C9_HUMAN | ||||||||
| KEGG Pathway | hsa:1559 | ||||||||
| Protein Family | Cytochrome P450 family | ||||||||
| Protein Function |
A cytochrome P450 monooxygenase involved in the metabolism of various endogenous substrates, including fatty acids and steroids (PubMed:7574697, PubMed:9866708, PubMed:9435160, PubMed:12865317, PubMed:15766564, PubMed:19965576, PubMed:21576599). Mechanistically, uses molecular oxygen inserting one oxygen atom into a substrate, and reducing the second into a water molecule, with two electrons provided by NADPH via cytochrome P450 reductase (NADPH--hemoprotein reductase) (PubMed:7574697, PubMed:9866708, PubMed:9435160, PubMed:12865317, PubMed:15766564, PubMed:19965576, PubMed:21576599). Catalyzes the epoxidation of double bonds of polyunsaturated fatty acids (PUFA) (PubMed:7574697, PubMed:15766564, PubMed:19965576, PubMed:9866708). Catalyzes the hydroxylation of carbon-hydrogen bonds. Metabolizes cholesterol toward 25-hydroxycholesterol, a physiological regulator of cellular cholesterol homeostasis (PubMed:21576599). Exhibits low catalytic activity for the formation of catechol estrogens from 17beta-estradiol (E2) and estrone (E1), namely 2-hydroxy E1 and E2 (PubMed:12865317). Catalyzes bisallylic hydroxylation and hydroxylation with double-bond migration of polyunsaturated fatty acids (PUFA) (PubMed:9866708, PubMed:9435160). Also metabolizes plant monoterpenes such as limonene. Oxygenates (R)- and (S)-limonene to produce carveol and perillyl alcohol (PubMed:11950794). Contributes to the wide pharmacokinetics variability of the metabolism of drugs such as S-warfarin, diclofenac, phenytoin, tolbutamide and losartan (PubMed:25994031).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised if sulfinpyrazone is used concomitantly with phenytoin. Dosage adjustments as well as clinical and laboratory monitoring should be considered whenever sulfinpyrazone is added to or withdrawn from therapy. Patients should be monitored for the development of phenytoin toxicity such as nausea, vomiting, tremors, ataxia, seizures, drowsiness, lethargy, slurred speech, visual disturbances, and changes in mental status. | ||||||||
