Details of Drug-Drug Interaction
| Drug General Information (ID: DDI1QY0FMK) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Dicoumarol | Drug Info | Cholestyramine | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Anticoagulants | Bile Acid Sequestrants | |||||||
| Structure | |||||||||
| Mechanism of Dicoumarol-Cholestyramine Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
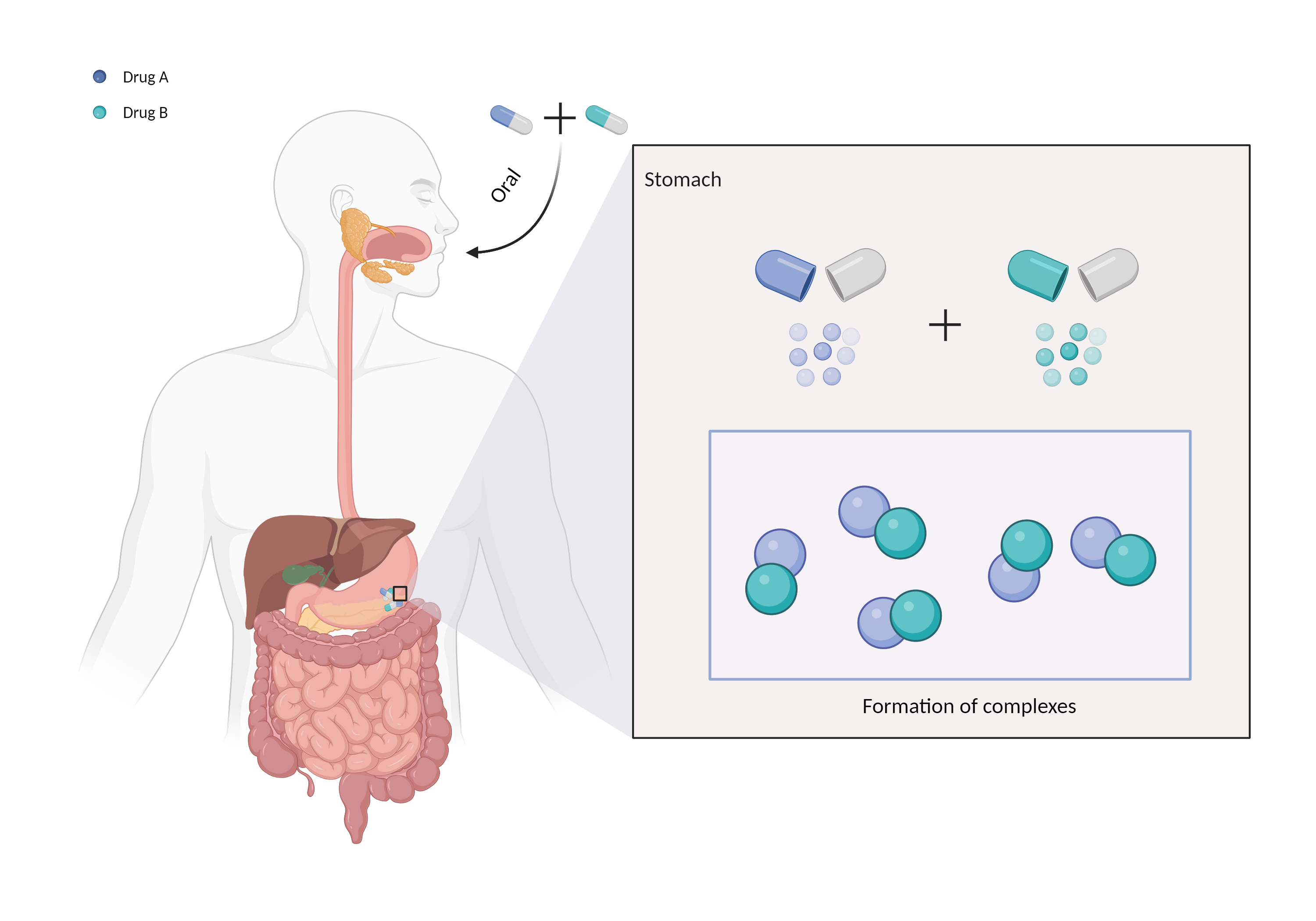
| Complex formation Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Dicoumarol | Cholestyramine | |||||||
| Mechanism | Binds to anion exchange resin/polymer | Anion exchange resin/polymer | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Chelation | ||||||||
| Factor Description | Chelation is a direct drug interaction that usually involves the formation of dimers or trimers, resulting in larger complexes and poorer absorption. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Warfarin or other oral anticoagulants should be administered at least one hour before or four to six hours after cholestyramine. The INR or PT should be closely monitored. Patients should be advised to promptly report any signs of bleeding (e.g., pain, swelling, headache, dizziness, weakness, vaginal bleeding, nosebleeds, bleeding of gums from brushing, unusual bleeding or bruising, red or brown urine, or red or black stools) or clots (e.g. chest pain, shortness of breath, sudden loss of vision, or pain, redness or swelling in an extremity). | ||||||||
