Details of Drug-Drug Interaction
| Drug General Information (ID: DDI0TVBHAO) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Didanosine | Drug Info | Hydroxyurea | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Anti-Hiv Agents | Antineoplastics | |||||||
| Structure | |||||||||
| Mechanism of Didanosine-Hydroxyurea Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
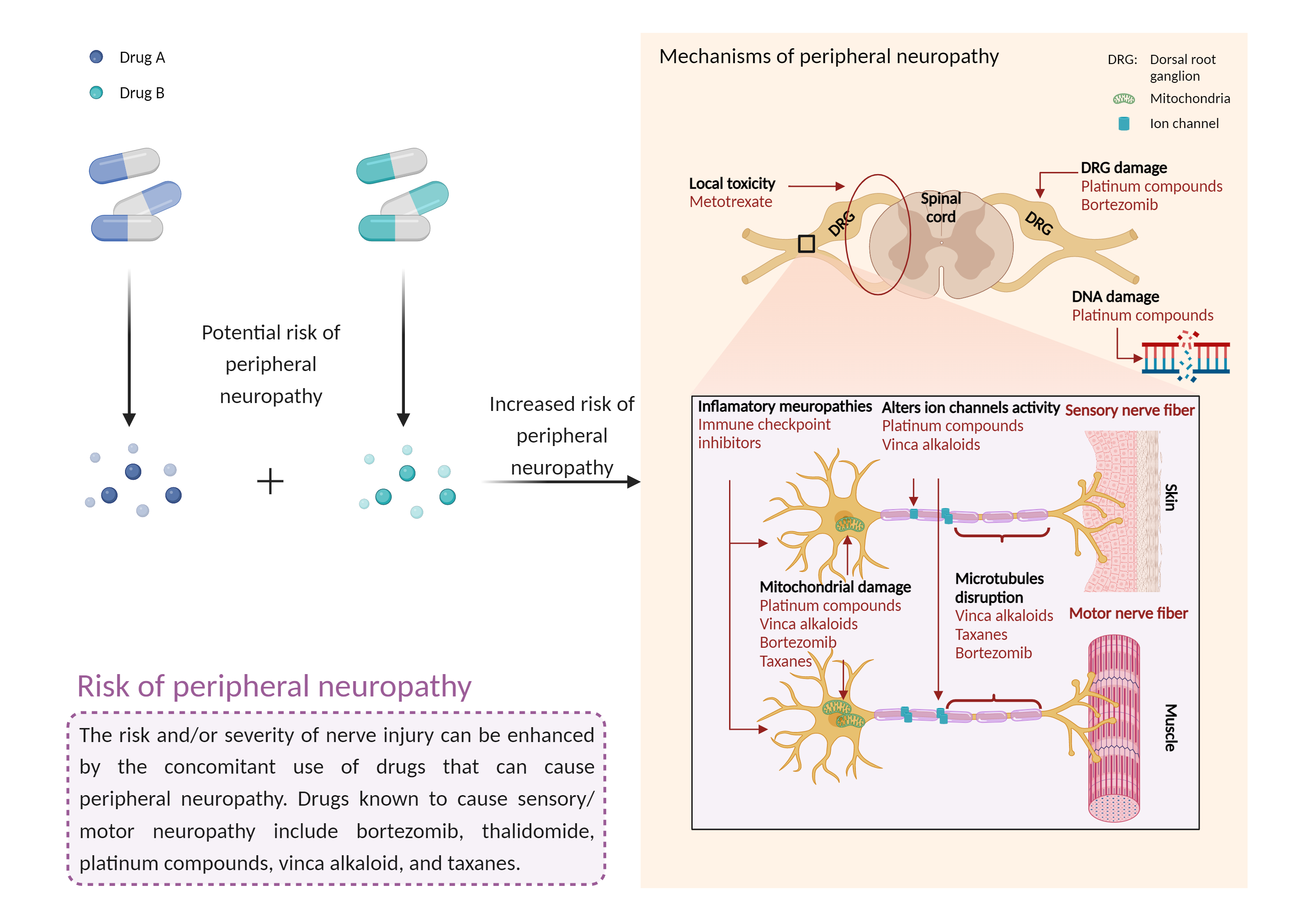
| Increased risk of peripheral neuropathy Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Didanosine | Hydroxyurea | |||||||
| Mechanism 1 | Peripheral neuropathy | Peripheral neuropathy | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Peripheral neuropathy | ||||||||
| Factor Description | Peripheral neuropathy is a disorder of the peripheral nerves that can cause sensory and motor symptoms, and even paralysis in severe cases. Signs and symptoms of peripheral neuropathy may include: gradual numbness, tingling or prickling in the feet or hands that can spread up the legs and arms; sharp, stinging, pricking or burning sensations; extreme sensitivity to touch; pain during activities that should not cause pain; lack of coordination and falls; muscle weakness; and paralysis if the motor nerves are affected. | ||||||||
| Mechanism Description |
|
||||||||
| Increased risk of other adverse reactions (Unspecific) Click to Show/Hide Mechanism Graph | |||||||||
| Drug Name | Didanosine | Hydroxyurea | |||||||
| Mechanism 2 | Pancreatic toxicity | Pancreatic toxicity | |||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Adverse reactions | ||||||||
| Factor Description | An adverse reaction is an unexpected negative reaction to a medication or treatment that happens even when it's used correctly. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | In general, antiretroviral regimens containing ddI and hydroxyurea should probably not be prescribed if other treatment options are available. Close observation for signs and symptoms of toxicity is recommended if these drugs must be used together, particularly in patients with advanced HIV disease and/or elderly patients. Both drugs should be suspended if pancreatitis is suspected, and ddI discontinued permanently if pancreatitis is confirmed. Prompt withdrawal of these drugs is also essential in patients who develop peripheral neuropathy to avoid irreversible damage. Therapy with ddI may be reinstituted following resolution of neuropathy symptoms, but reduced dosages are recommended. Likewise, treatment should be suspended in any patient who develops clinical or laboratory findings suggestive of lactic acidosis or pronounced hepatotoxicity, and permanent discontinuation considered if the syndrome is confirmed. Patients should be advised to seek medical attention promptly if symptoms of toxicity occur such as nausea, vomiting, abdominal pain, fatigue, unexplained weight loss, tachypnea, dyspnea, motor weakness, numbness, tingling, and pain in hands and feet. | ||||||||
