Details of Drug-Drug Interaction
| Drug General Information (ID: DDI0R7YIE8) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Bumetanide | Drug Info | Etidronic acid | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Diuretics | Bone Density Conservation Agents | |||||||
| Structure | |||||||||
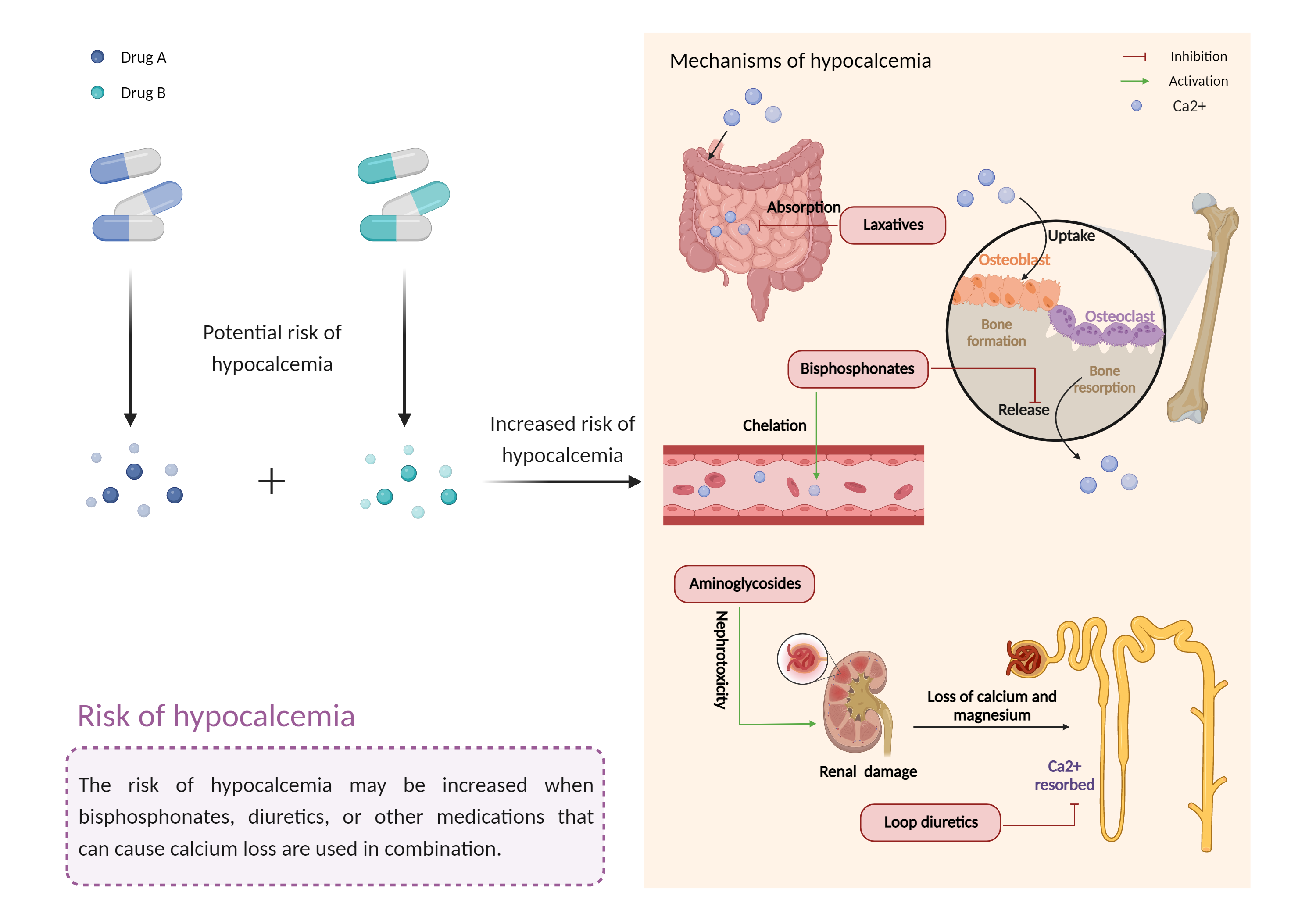
| Mechanism of Bumetanide-Etidronic acid Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Increased risk of hypocalcemia Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Bumetanide | Etidronic acid | |||||||
| Mechanism | Hypocalcemia | Hypocalcemia | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Hypocalcemia | ||||||||
| Factor Description | Hypocalcemia is a low level of calcium found in a blood test. It can cause symptoms such as tingling, muscle cramps and heart rhythm problems that can range from mild to life-threatening. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised if bisphosphonates are coadministered with loop diuretics. Renal function as well as serum calcium levels should be monitored during and after treatment, since the effects of bisphosphonates can continue for many weeks after discontinuation. Patients and clinicians should be alert to signs and symptoms of clinical hypocalcemia such as muscle spasm, carpopedal spasm, laryngeal spasm, tetany, convulsions, mental changes (irritability, depression, psychosis), increased intracranial pressure, papilledema, QT interval prolongation, and cardiac arrhythmias. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Skelid (tilundronate). Sanofi Winthrop Pharmaceuticals, New York, NY. | ||||||||||||||||||
| 2 | Product Information. Zometa (zoledronic acid). Novartis Pharmaceuticals, East Hanover, NJ. | ||||||||||||||||||
