Details of Drug-Drug Interaction
| Drug General Information (ID: DDI0DPOITA) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Cisapride | Drug Info | Sirolimus | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Gastrointestinal Agents | Immunosuppressive Agents | |||||||
| Structure | |||||||||
| Mechanism of Cisapride-Sirolimus Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
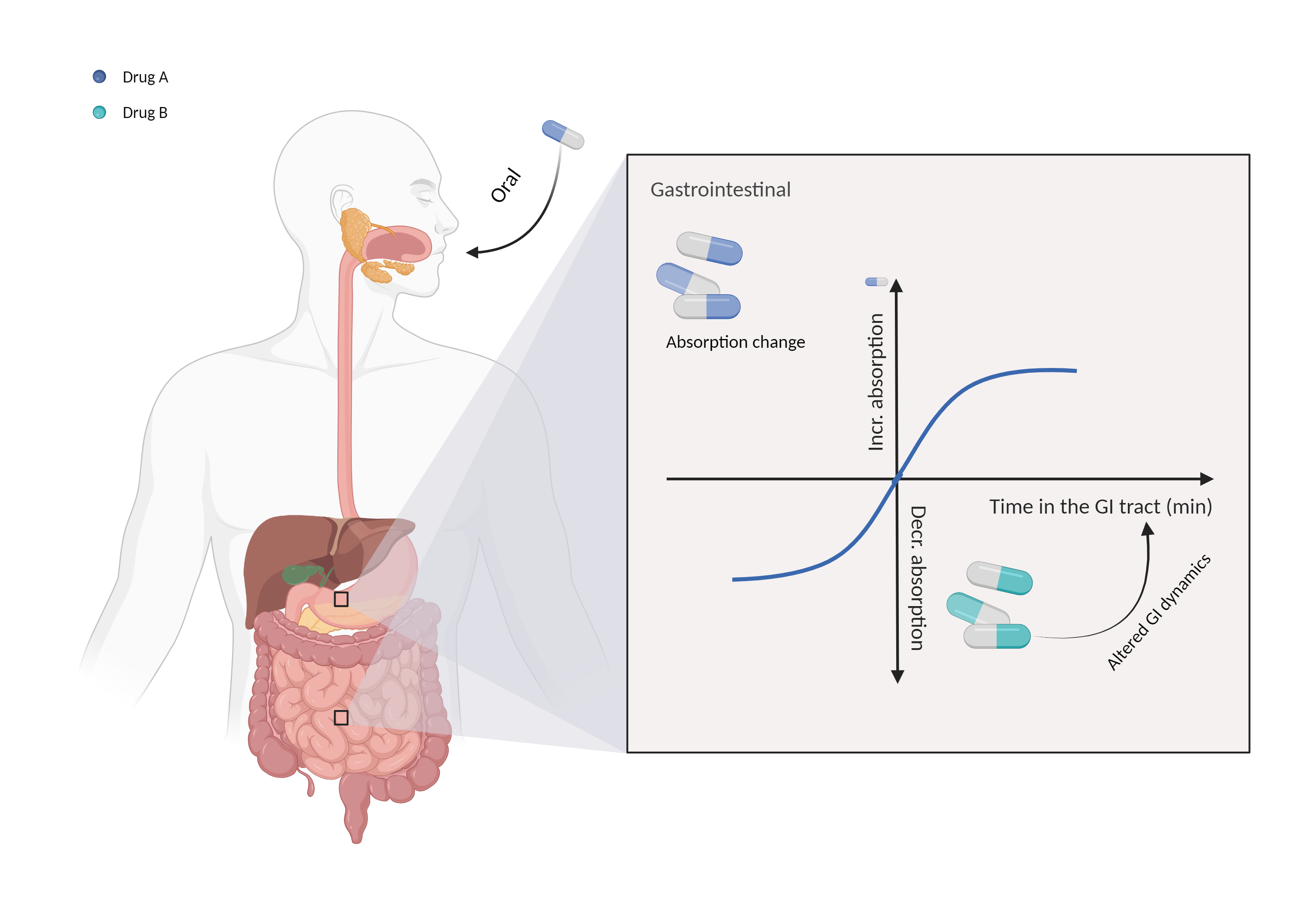
| Altered gastrointestinal dynamics Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Cisapride | Sirolimus | |||||||
| Mechanism | Increase gastrointestinal motility | Absorption is influenced by gastrointestinal motility | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Gastrointestinal Motility | ||||||||
| Factor Description | Gastrointestinal motility is an important factor in determining the absorption of orally administered drugs, and it controls the residence time of the drug in the digestive tract. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Pharmacologic response, renal function, and macrolide immunosuppressant blood levels should be monitored more closely whenever a gastrointestinal prokinetic agent is added to or discontinue from therapy, and the immunosuppressant dosage adjusted as necessary. Patients should be advised to contact their physician if they experience potential signs and symptoms of macrolide immunosuppressant toxicity such as fever, infection, diarrhea, tremor, headache, fatigue, or lethargy. | ||||||||
