Details of Drug-Drug Interaction
| Drug General Information (ID: DDI0C7RJO8) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Tetrabenazine | Drug Info | Amisulpride | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Nephropathic Cystinosis Therapy | Antipsychotic Agents | |||||||
| Structure | |||||||||
| Mechanism of Tetrabenazine-Amisulpride Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
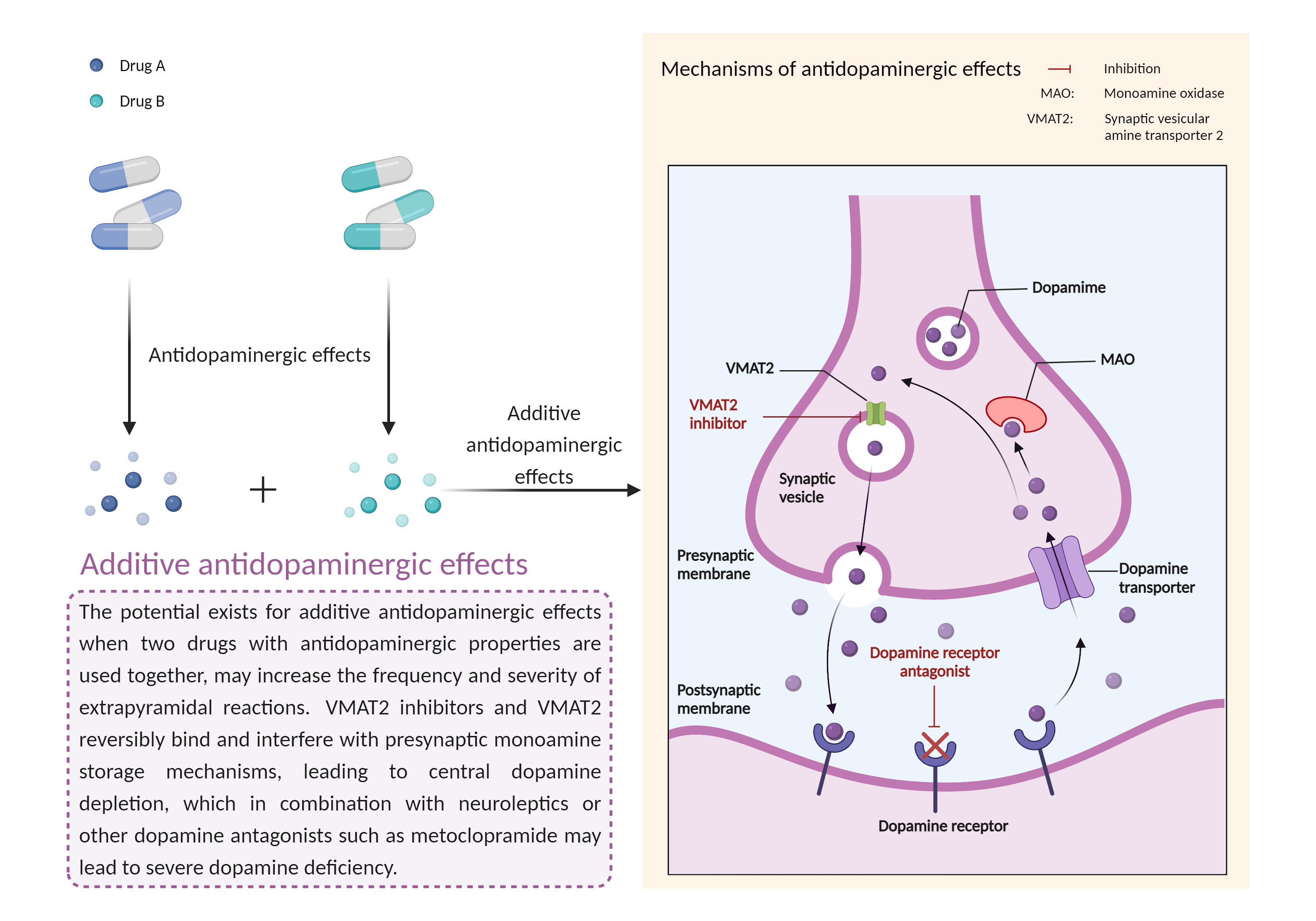
| Additive antidopaminergic effects Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Tetrabenazine | Amisulpride | |||||||
| Mechanism 1 |
Antidopaminergic effects Synaptic vesicular amine transporter Inhibitor |
Antidopaminergic effects Dopamine receptor Antagonist |
|||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Synaptic vesicle amine transporter |
×
Structure
Sequence
MALSELALVRWLQESRRSRKLILFIVFLALLLDNMLLTVVVPIIPSYLYSIKHEKNATEIQTARPVHTASISDSFQSIFSYYDNSTMVTGNATRDLTLHQTATQHMVTNASAVPSDCPSEDKDLLNENVQVGLLFASKATVQLITNPFIGLLTNRIGYPIPIFAGFCIMFVSTIMFAFSSSYAFLLIARSLQGIGSSCSSVAGMGMLASVYTDDEERGNVMGIALGGLAMGVLVGPPFGSVLYEFVGKTAPFLVLAALVLLDGAIQLFVLQPSRVQPESQKGTPLTTLLKDPYILIAAGSICFANMGIAMLEPALPIWMMETMCSRKWQLGVAFLPASISYLIGTNIFGILAHKMGRWLCALLGMIIVGVSILCIPFAKNIYGLIAPNFGVGFAIGMVDSSMMPIMGYLVDLRHVSVYGSVYAIADVAFCMGYAIGPSAGGAIAKAIGFPWLMTIIGIIDILFAPLCFFLRSPPAKEEKMAILMDHNCPIKTKMYTQNNIQSYPIGEDEESESD
|
|||||||
| Gene Name | SLC18A2 | ||||||||
| Uniprot ID | VMAT2_HUMAN | ||||||||
| KEGG Pathway | hsa:6571 | ||||||||
| Protein Family | Major facilitator superfamily | ||||||||
| Protein Function |
Involved in the ATP-dependent vesicular transport of biogenic amine neurotransmitters. Pumps cytosolic monoamines including dopamine, norepinephrine, serotonin, and histamine into synaptic vesicles (PubMed:23363473). Requisite for vesicular amine storage prior to secretion via exocytosis.
Click to Show/Hide
|
||||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Dopamine receptor | Structure Sequence | |||||||
| Protein Family | G-protein coupled receptor 1 family | ||||||||
| Protein Function |
Dopamine receptor whose activity is mediated by G proteins which activate adenylyl cyclase.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Increased risk of prolong QT interval Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Tetrabenazine | Amisulpride | |||||||
| Mechanism 2 | Prolong QT interval | Prolong QT interval | |||||||
| Key Mechanism Factor 3 | |||||||||
| Factor Name | QT interval | ||||||||
| Factor Description | Long QT syndrome is a heart signaling disorder that can cause a fast, chaotic heartbeat (arrhythmia). Many people may not exhibit symptoms, and usually the condition is detected during routine medical tests. In others, the most common symptoms include: sudden fainting, palpitations, dizziness, seizures, sudden death. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | The use of tetrabenazine in combination with neuroleptic agents or other dopamine antagonists should preferably be avoided. When coadministration is required, patients should be instructed to notify their physician if they experience extrapyramidal symptoms such as bradykinesia, hypertonia, rigidity, restlessness, and dysphagia. Clinicians, caregivers, and family members should be apprised of the risk of neuroleptic malignant syndrome and be alert to potential signs and symptoms such as mental status changes (e.g., mutism, catatonia, stupor, coma, agitation, confusion, hallucinations, delusions), autonomic instability, restlessness, rigidity, ataxia, myoclonus, hyperreflexia, tremors, diaphoresis, elevated creatine phosphokinase levels, and hyperpyrexia. Patients should seek medical attention if they experience symptoms that could indicate the occurrence of torsade de pointes such as dizziness, palpitations, or syncope. | ||||||||
