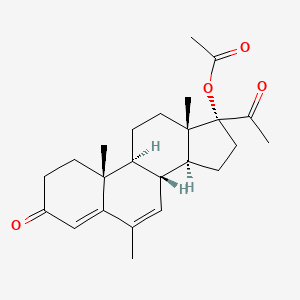
Details of Drug
| Drug-Drug Interaction Network | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Sunburst Graph |
AM001 Affected gastrointestinal absorption
AM002 Affected cellular transport
AM004 Affected intra/extra-hepatic metabolism
AM006 Pharmacodynamic additive effects
AM007 Pharmacodynamic antagonistic effects
BM003 Complex formation
BM010 Transporter inhibition
BM017 CYP450 enzyme inhibition
BM022 CYP450 enzyme induction
BM023 UGT induction
BM024 Competitive inhibition of metabolic enzyme
BM047 Additive thrombogenic effects
BM086 Antagonize the effect of antidiabetic agents
BM089 Antagonize the effect of antihypertensive agents
|
||||||||
| Relation Graph | |||||||||
| Full list of drugs interacting with Megestrol acetate | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Affected gastrointestinal absorption | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Complex formation | Drug Num: 2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0329
|
Cholestyramine
|
C27H47ClN2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0382
|
Colestipol
|
C11H28ClN5O
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Affected cellular transport | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transporter inhibition | Drug Num: 2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0513
|
Dofetilide
|
C19H27N3O5S2
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0412
|
Dalfampridine
|
C5H6N2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Affected intra/extra-hepatic metabolism | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CYP450 enzyme inhibition | Drug Num: 29 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0597
|
Erythromycin
|
C37H67NO13
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0002
|
Abametapir (topical)
|
C12H12N2
|
|
Moderate | Inter Info |
[19]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0100
|
Aprepitant
|
C23H21F7N4O3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0171
|
Berotralstat
|
C30H26F4N6O
|
|
Moderate | Inter Info |
[22]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0192
|
Boceprevir
|
C27H45N5O5
|
|
Moderate | Inter Info |
[23]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0349
|
Clarithromycin
|
C38H69NO13
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0386
|
Conivaptan
|
C32H26N4O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0426
|
Darunavir
|
C27H37N3O7S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0432
|
Deferasirox
|
C21H15N3O4
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0534
|
Dronedarone
|
C31H44N2O5S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0543
|
Duvelisib
|
C22H17ClN6O
|
|
Moderate | Inter Info |
[38]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0638
|
Fedratinib
|
C27H36N6O3S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0693
|
Fosaprepitant
|
C23H22F7N4O6P
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0699
|
Fostamatinib
|
C23H26FN6O9P
|
|
Moderate | Inter Info |
[44]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0852
|
Isavuconazonium
|
C35H35F2N8O5S+
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0869
|
Ivacaftor
|
C24H28N2O3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0894
|
Lapatinib
|
C29H26ClFN4O4S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0896
|
Larotrectinib
|
C21H22F2N6O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0899
|
Lefamulin
|
C28H45NO5S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0906
|
Letermovir
|
C29H28F4N4O4
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0946
|
Lonafarnib
|
C27H31Br2ClN4O2
|
|
Moderate | Inter Info |
[54]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1063
|
Mifepristone
|
C29H35NO2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1136
|
Nilotinib
|
C28H22F3N7O
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1315
|
Posaconazole
|
C37H42F2N8O4
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1432
|
Rucaparib
|
C19H18FN3O
|
|
Moderate | Inter Info |
[61]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1459
|
Selpercatinib
|
C29H31N7O3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1541
|
Telaprevir
|
C36H53N7O6
|
|
Moderate | Inter Info |
[67]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1544
|
Telithromycin
|
C43H65N5O10
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1706
|
Voriconazole
|
C16H14F3N5O
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CYP450 enzyme induction | Drug Num: 49 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0178
|
Bexarotene
|
C24H28O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0224
|
Butabarbital
|
C10H16N2O3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0225
|
Butalbital
|
C11H16N2O3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0061
|
Aminoglutethimide
|
C13H16N2O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0071
|
Amobarbital
|
C11H18N2O3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0095
|
Apalutamide
|
C21H15F4N5O2S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0194
|
Bosentan
|
C27H29N5O6S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0204
|
Brigatinib
|
C29H39ClN7O2P
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0255
|
Carbamazepine
|
C15H12N2O
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0406
|
Dabrafenib
|
C23H20F3N5O2S2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0545
|
Echinacea
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0554
|
Efavirenz
|
C14H9ClF3NO2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0555
|
Elagolix
|
C32H30F5N3O5
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0578
|
Enzalutamide
|
C21H16F4N4O2S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0601
|
Eslicarbazepine
|
C15H14N2O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0639
|
Felbamate
|
C11H14N2O4
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0697
|
Fosphenytoin
|
C16H15N2O6P
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0741
|
Griseofulvin
|
C17H17ClO6
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0872
|
Ivosidenib
|
C28H22ClF3N6O3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0901
|
Lemborexant
|
C22H20F2N4O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0953
|
Lorlatinib
|
C21H19FN6O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0958
|
Lumacaftor
|
C24H18F2N2O5
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1040
|
Methylphenobarbital
|
C13H14N2O3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1078
|
Mitotane
|
C14H10Cl4
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1099
|
Nafcillin
|
C21H22N2O5S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1198
|
Oxcarbazepine
|
C15H12N2O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1258
|
Pexidartinib
|
C20H15ClF3N5
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1265
|
Phenobarbital
|
C12H12N2O3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1274
|
Phenylbutazone
|
C19H20N2O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1340
|
Primidone
|
C12H14N2O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1399
|
Rifabutin
|
C46H62N4O11
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1400
|
Rifampicin
|
C43H58N4O12
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1401
|
Rifapentine
|
C47H64N4O12
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1433
|
Rufinamide
|
C10H8F2N4O
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1453
|
Secobarbital
|
C12H18N2O3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1493
|
Somapacitan
|
C54H95N13O20S2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1495
|
Somatrem
|
C43H72N14O14
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1502
|
St. John's Wort
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1518
|
Sulfinpyrazone
|
C23H20N2O3S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1538
|
Tazemetostat
|
C34H44N4O4
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1546
|
Telotristat ethyl
|
C27H26ClF3N6O3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1687
|
Vemurafenib
|
C23H18ClF2N3O3S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1128
|
Nevirapine
|
C15H14N4O
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1248
|
Pentobarbital
|
C11H18N2O3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1283
|
Phenytoin
|
C15H12N2O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1494
|
Somatotropin
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1652
|
Troglitazone
|
C24H27NO5S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0106
|
Armodafinil
|
C15H15NO2S
|
|
Minor | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1081
|
Modafinil
|
C15H15NO2S
|
|
Minor | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| UGT induction | Drug Num: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1677
|
Valproic acid
|
C8H16O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Competitive inhibition of metabolic enzyme | Drug Num: 3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0402
|
Cyclosporine
|
C62H111N11O12
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0696
|
Fosnetupitant
|
C31H35F6N4O5P
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1127
|
Netupitant
|
C30H32F6N4O
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pharmacodynamic additive effects | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Additive thrombogenic effects | Drug Num: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1569
|
Thalidomide
|
C13H10N2O4
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pharmacodynamic antagonistic effects | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Antagonize the effect of antidiabetic agents | Drug Num: 39 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0009
|
Acarbose
|
C25H43NO18
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0727
|
Glimepiride
|
C24H34N4O5S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0811
|
Insulin glargine
|
C267H404N72O78S6
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0813
|
Insulin human
|
C257H383N65O77S6
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0814
|
Insulin human (inhalation, rapid acting)
|
C257H383N65O77S6
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0815
|
Insulin human (isophane)
|
C257H383N65O77S6
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0816
|
Insulin human (regular)
|
C257H383N65O77S6
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0817
|
Insulin human (zinc extended)
|
C257H383N65O77S6
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0818
|
Insulin human (zinc)
|
C257H383N65O77S6
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0819
|
Insulin lispro
|
C257H389N65O77S6
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0820
|
Insulin lispro (protamine)
|
C257H389N65O77S6
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0013
|
Acetohexamide
|
C15H20N2O4S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0029
|
Albiglutide
|
C148H224N40O45
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0042
|
Alogliptin
|
C18H21N5O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0245
|
Canagliflozin
|
C24H25FO5S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0325
|
Chlorpropamide
|
C10H13ClN2O3S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0418
|
Dapagliflozin
|
C21H25ClO6
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0565
|
Empagliflozin
|
C23H27ClO7
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0728
|
Glipizide
|
C21H27N5O4S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0730
|
Glyburide
|
C23H28ClN3O5S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0807
|
Insulin aspart (aspart protamine)
|
C256H381N65O79S6
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0808
|
Insulin aspart (aspart)
|
C256H381N65O79S6
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0809
|
Insulin degludec
|
C274H411N65O81S6
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0810
|
Insulin detemir
|
C267H402N64O76S6
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0812
|
Insulin glulisine
|
C258H384N64O78S6
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0930
|
Linagliptin
|
C25H28N8O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0936
|
Liraglutide
|
C172H265N43O51
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0941
|
Lixisenatide
|
C215H347N61O65S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1064
|
Miglitol
|
C8H17NO5
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1112
|
Nateglinide
|
C19H27NO3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1294
|
Pioglitazone
|
C19H20N2O3S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1326
|
Pramlintide
|
C171H267N51O53S2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1392
|
Repaglinide
|
C27H36N2O4
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1429
|
Rosiglitazone
|
C18H19N3O3S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1449
|
Saxagliptin
|
C18H25N3O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1477
|
Sitagliptin
|
C16H15F6N5O
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1601
|
Tolazamide
|
C14H21N3O3S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1603
|
Tolbutamide
|
C12H18N2O3S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1015
|
Metformin
|
C4H11N5
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Antagonize the effect of antihypertensive agents | Drug Num: 4 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1779
|
Dulaglutide
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1782
|
Ertugliflozin
|
C22H25ClO7
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1784
|
Exenatide
|
C184H282N50O60S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1887
|
Semaglutide
|
C187H291N45O59
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||