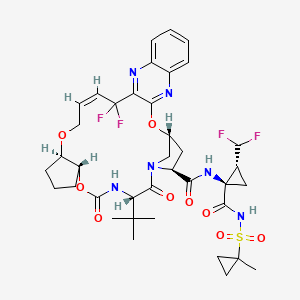
Details of Drug
| Drug-Drug Interaction Network | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Sunburst Graph |
AM002 Affected cellular transport
AM004 Affected intra/extra-hepatic metabolism
AM006 Pharmacodynamic additive effects
BM010 Transporter inhibition
BM011 Transporter induction
BM017 CYP450 enzyme inhibition
BM019 UGT inhibition
BM022 CYP450 enzyme induction
BM109 Increased risk of other adverse reactions (Unspecific)
|
||||||||
| Relation Graph | |||||||||
| Full list of drugs interacting with Glecaprevir | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Affected cellular transport | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transporter inhibition | Drug Num: 46 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0116
|
Atazanavir
|
C38H52N6O7
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0380
|
Colchicine
|
C22H25NO6
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0402
|
Cyclosporine
|
C62H111N11O12
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0426
|
Darunavir
|
C27H37N3O7S
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0551
|
Edoxaban
|
C24H30ClN7O4S
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0171
|
Berotralstat
|
C30H26F4N6O
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0956
|
Lovastatin
|
C24H36O5
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1232
|
Pazopanib
|
C21H23N7O2S
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1329
|
Pravastatin
|
C23H36O7
|
|
Major | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1388
|
Relugolix
|
C29H27F2N7O5S
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1430
|
Rosuvastatin
|
C22H28FN3O6S
|
|
Major | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1688
|
Venetoclax
|
C45H50ClN7O7S
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0038
|
Aliskiren
|
C30H53N3O6
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0096
|
Apixaban
|
C25H25N5O4
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0155
|
Bempedoic acid
|
C19H36O5
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0176
|
Betrixaban
|
C23H22ClN5O3
|
|
Moderate | Inter Info |
[30]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0182
|
Binimetinib
|
C17H15BrF2N4O3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0195
|
Bosutinib
|
C26H29Cl2N5O3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0199
|
Brentuximab vedotin
|
NA
|
NA
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0375
|
Cobicistat
|
C40H53N7O5S2
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0376
|
Cobimetinib
|
C21H21F3IN3O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0425
|
Darolutamide
|
C19H19ClN6O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0429
|
Daunorubicin
|
C27H29NO10
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0430
|
Daunorubicin (liposomal)
|
C27H29NO10
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0491
|
Digoxin
|
C41H64O14
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0568
|
Enasidenib
|
C19H17F6N7O
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0577
|
Entrectinib
|
C31H34F2N6O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0629
|
Everolimus
|
C53H83NO14
|
|
Moderate | Inter Info |
[42]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0686
|
Fluvastatin
|
C24H26FNO4
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0720
|
Gilteritinib
|
C29H44N8O3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0896
|
Larotrectinib
|
C21H22F2N6O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1409
|
Ripretinib
|
C24H21BrFN5O2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1663
|
Ubrogepant
|
C29H26F3N5O3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0788
|
Idarubicin
|
C26H27NO9
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0789
|
Idelalisib
|
C22H18FN7O
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0867
|
Itraconazole
|
C35H38Cl2N8O4
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0899
|
Lefamulin
|
C28H45NO5S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0963
|
Lusutrombopag
|
C29H32Cl2N2O5S
|
|
Moderate | Inter Info |
[51]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0980
|
Maraviroc
|
C29H41F2N5O
|
|
Moderate | Inter Info |
[52]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1101
|
Naldemedine
|
C32H34N4O6
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1301
|
Pitavastatin
|
C25H24FNO4
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1396
|
Revefenacin
|
C35H43N5O4
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1532
|
Talazoparib
|
C19H14F2N6O
|
|
Moderate | Inter Info |
[57]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0658
|
Fidaxomicin
|
C52H74Cl2O18
|
|
Minor | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0797
|
Indacaterol
|
C24H28N2O3
|
|
Minor | Inter Info |
[60]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1104
|
Naloxegol
|
C34H53NO11
|
|
Minor | Inter Info |
[61]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transporter induction | Drug Num: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1592
|
Tipranavir
|
C31H33F3N2O5S
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Affected intra/extra-hepatic metabolism | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CYP450 enzyme inhibition | Drug Num: 5 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0120
|
Atorvastatin
|
C33H35FN2O5
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1473
|
Simvastatin
|
C25H38O5
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1476
|
Sirolimus
|
C51H79NO13
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1526
|
Tacrolimus
|
C44H69NO12
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1549
|
Temsirolimus
|
C56H87NO16
|
|
Moderate | Inter Info |
[58]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CYP450 enzyme induction | Drug Num: 47 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0255
|
Carbamazepine
|
C15H12N2O
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0554
|
Efavirenz
|
C14H9ClF3NO2
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1265
|
Phenobarbital
|
C12H12N2O3
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1283
|
Phenytoin
|
C15H12N2O2
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1340
|
Primidone
|
C12H14N2O2
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1400
|
Rifampicin
|
C43H58N4O12
|
|
Major | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1502
|
St. John's Wort
|
NA
|
NA
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0061
|
Aminoglutethimide
|
C13H16N2O2
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0071
|
Amobarbital
|
C11H18N2O3
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0095
|
Apalutamide
|
C21H15F4N5O2S
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0106
|
Armodafinil
|
C15H15NO2S
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0178
|
Bexarotene
|
C24H28O2
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0194
|
Bosentan
|
C27H29N5O6S
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0204
|
Brigatinib
|
C29H39ClN7O2P
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0224
|
Butabarbital
|
C10H16N2O3
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0225
|
Butalbital
|
C11H16N2O3
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0300
|
Cenobamate
|
C10H10ClN5O2
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0432
|
Deferasirox
|
C21H15N3O4
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0453
|
Dexamethasone
|
C22H29FO5
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0555
|
Elagolix
|
C32H30F5N3O5
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0578
|
Enzalutamide
|
C21H16F4N4O2S
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0628
|
Etravirine
|
C20H15BrN6O
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0639
|
Felbamate
|
C11H14N2O4
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0697
|
Fosphenytoin
|
C16H15N2O6P
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0601
|
Eslicarbazepine
|
C15H14N2O2
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0872
|
Ivosidenib
|
C28H22ClF3N6O3
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0741
|
Griseofulvin
|
C17H17ClO6
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0901
|
Lemborexant
|
C22H20F2N4O2
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0905
|
Lesinurad
|
C17H14BrN3O2S
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0953
|
Lorlatinib
|
C21H19FN6O2
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1040
|
Methylphenobarbital
|
C13H14N2O3
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1078
|
Mitotane
|
C14H10Cl4
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1099
|
Nafcillin
|
C21H22N2O5S
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1128
|
Nevirapine
|
C15H14N4O
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1186
|
Oritavancin
|
C86H97Cl3N10O26
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1190
|
Osimertinib
|
C28H33N7O2
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1198
|
Oxcarbazepine
|
C15H12N2O2
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1248
|
Pentobarbital
|
C11H18N2O3
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1258
|
Pexidartinib
|
C20H15ClF3N5
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1274
|
Phenylbutazone
|
C19H20N2O2
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1399
|
Rifabutin
|
C46H62N4O11
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1401
|
Rifapentine
|
C47H64N4O12
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1433
|
Rufinamide
|
C10H8F2N4O
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1453
|
Secobarbital
|
C12H18N2O3
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1518
|
Sulfinpyrazone
|
C23H20N2O3S
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1652
|
Troglitazone
|
C24H27NO5S
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1687
|
Vemurafenib
|
C23H18ClF2N3O3S
|
|
Moderate | Inter Info |
[1]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pharmacodynamic additive effects | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Increased risk of other adverse reactions (Unspecific) | Drug Num: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0619
|
Ethinylestradiol
|
C20H24O2
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||