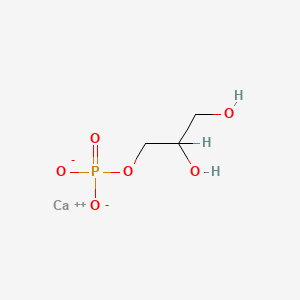
Details of Drug
| Drug-Drug Interaction Network | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Sunburst Graph |
AM001 Affected gastrointestinal absorption
AM006 Pharmacodynamic additive effects
AM007 Pharmacodynamic antagonistic effects
BM003 Complex formation
BM089 Antagonize the effect of antihypertensive agents
BM107 Attenuated pharmacological effects (Unspecific)
BM108 Increased risk of hypercalcemia
BM109 Increased risk of other adverse reactions (Unspecific)
|
||||||||
| Relation Graph | |||||||||
| Full list of drugs interacting with Calcium glycerophosphate | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Affected gastrointestinal absorption | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Complex formation | Drug Num: 18 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0181
|
Bictegravir
|
C21H18F3N3O5
|
|
Major | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0515
|
Dolutegravir
|
C20H19F2N3O5
|
|
Major | Inter Info |
[6]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0034
|
Alendronic acid
|
C4H13NO7P2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0144
|
Baloxavir marboxil
|
C27H23F2N3O7S
|
|
Moderate | Inter Info |
[15]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0438
|
Delafloxacin
|
C18H12ClF3N4O4
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0341
|
Ciprofloxacin
|
C17H18FN3O3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0433
|
Deferiprone
|
C7H9NO2
|
|
Moderate | Inter Info |
[51]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0561
|
Eltrombopag
|
C25H22N4O4
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0623
|
Etidronic acid
|
C2H8O7P2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0712
|
Gatifloxacin
|
C19H22FN3O4
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0716
|
Gemifloxacin
|
C18H20FN5O4
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0919
|
Levofloxacin
|
C18H20FN3O4
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1092
|
Moxifloxacin
|
C21H24FN3O4
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1155
|
Norfloxacin
|
C16H18FN3O3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1165
|
Ofloxacin
|
C18H20FN3O4
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1245
|
Penicillamine
|
C5H11NO2S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1412
|
Risedronic acid
|
C7H11NO7P2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1657
|
Trovafloxacin
|
C20H15F3N4O3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pharmacodynamic additive effects | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Increased risk of hypercalcemia | Drug Num: 9 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1753
|
Calcipotriol (topical)
|
C27H40O3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1754
|
Calcitriol (topical)
|
C27H44O3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0321
|
Chlorothiazide
|
C7H6ClN3O4S2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0326
|
Chlorthalidone
|
C14H11ClN2O4S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0771
|
Hydrochlorothiazide
|
C7H8ClN3O4S2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0775
|
Hydroflumethiazide
|
C8H8F3N3O4S2
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0798
|
Indapamide
|
C16H16ClN3O3S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1048
|
Metolazone
|
C16H16ClN3O3S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1311
|
Polythiazide
|
C11H13ClF3N3O4S3
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Increased risk of other adverse reactions (Unspecific) | Drug Num: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0221
|
Burosumab
|
NA
|
NA
|
Major | Inter Info |
[4]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pharmacodynamic antagonistic effects | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Antagonize the effect of antihypertensive agents | Drug Num: 10 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0069
|
Amlodipine
|
C20H25ClN2O5
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0497
|
Diltiazem
|
C22H26N2O4S
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0640
|
Felodipine
|
C18H19Cl2NO4
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0865
|
Isradipine
|
C19H21N3O5
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0911
|
Levamlodipine
|
C20H25ClN2O5
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1130
|
Nicardipine
|
C26H29N3O6
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1134
|
Nifedipine
|
C17H18N2O6
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1138
|
Nimodipine
|
C21H26N2O7
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1141
|
Nisoldipine
|
C20H24N2O6
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D1690
|
Verapamil
|
C27H38N2O4
|
|
Moderate | Inter Info | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Attenuated pharmacological effects (Unspecific) | Drug Num: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Drug ID
|
Drug Name
|
Formula | Pubchem ID | Severity Level | Interaction Detail | REF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
D0590
|
Erdafitinib
|
C25H30N6O2
|
|
Major | Inter Info |
[5]
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||