Details of Drug-Drug Interaction
| Drug General Information (ID: DDIZSUI687) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Ranitidine (bismuth citrate) | Drug Info | Dacomitinib | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antiulcer Agents | Antineoplastics/Her2 Inhibitors | |||||||
| Structure | |||||||||
| Mechanism of Ranitidine (bismuth citrate)-Dacomitinib Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
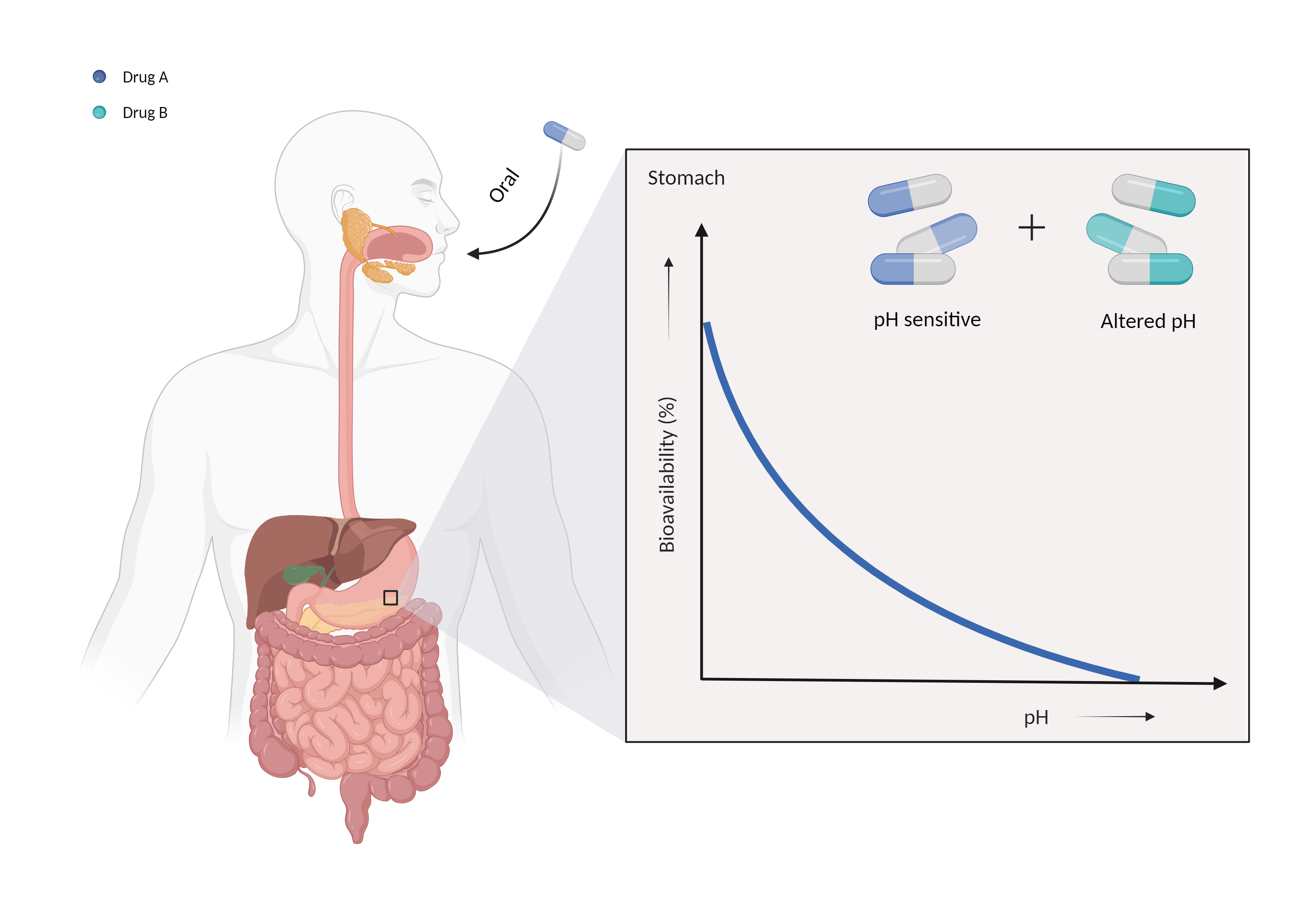
| Altered gastric pH Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Ranitidine (bismuth citrate) | Dacomitinib | |||||||
| Mechanism | Gastric alkalinizer | Gastric pH sensitive | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Gastric pH | ||||||||
| Factor Description | The normal pH range of gastric acid is between 1.5 and 3.5 and is highly acidic, consisting mainly of hydrochloric acid. Changes in the pH of the stomach can alter the absorption of drugs. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | The manufacturer recommends taking dacomitinib at least 6 hours before or 10 hours after H2-receptor antagonists. As an alternative, locally-acting antacids may be considered. Concomitant use of dacomitinib with PPIs should generally be avoided. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Cerner Multum, Inc. "UK Summary of Product Characteristics.". | ||||||||||||||||||
| 2 | Product Information. Vizimpro (dacomitinib). Pfizer U.S. Pharmaceuticals Group, New York, NY. | ||||||||||||||||||
