| Mechanism of Tacrolimus-Valganciclovir Interaction
(Severity Level: Moderate)
|
|
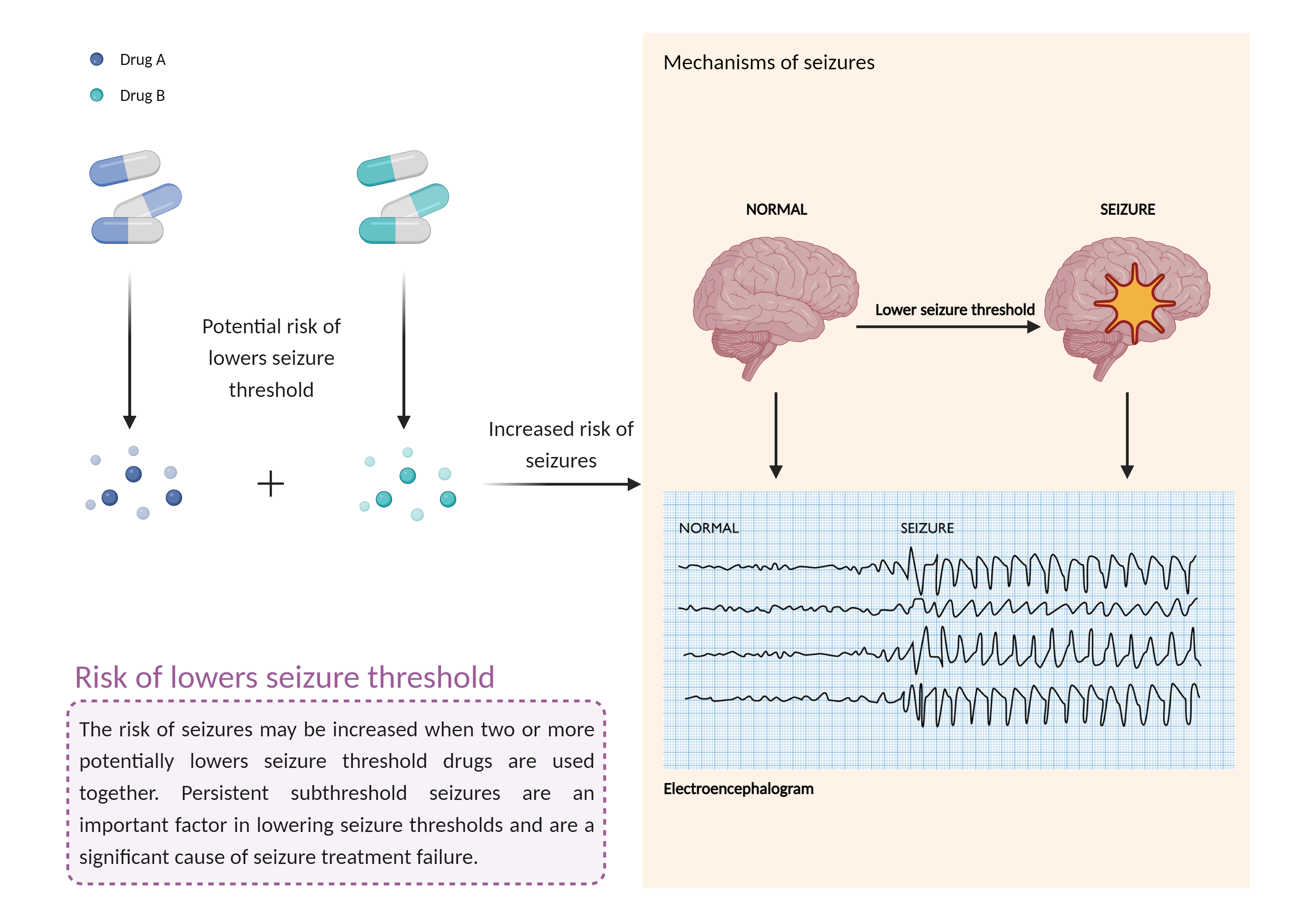
Increased risk of lowers seizure threshold
Click to Show/Hide Mechanism Graph
|
 |
| Drug Name |
Tacrolimus |
Valganciclovir |
|
Mechanism 1
|
Lower seizure threshold |
Lower seizure threshold |
| Key Mechanism Factor 1 |
| Factor Name |
Lowers seizure threshold |
| Factor Description |
The combination of medications that lower the seizure threshold is a factor that makes people with epilepsy more likely to have seizures. A seizure is a sudden, uncontrolled electrical disturbance in the brain that can cause changes in your behavior, movements or sensations, and level of consciousness. |
| Mechanism Description |
- Increased risk of lowers seizure threshold by the combination of Tacrolimus and Valganciclovir
|
|
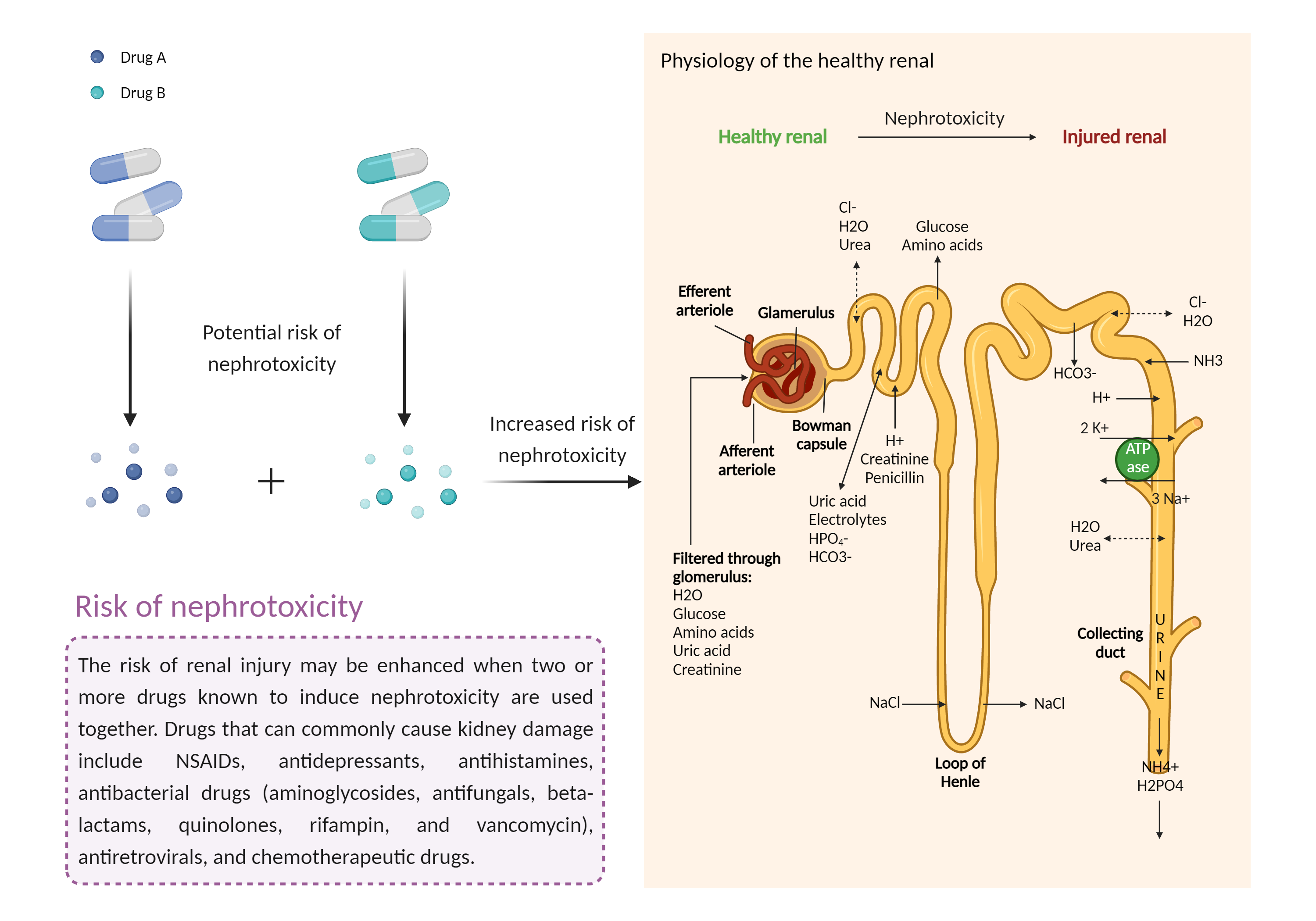
Increased risk of nephrotoxicity
Click to Show/Hide Mechanism Graph
|
 |
| Drug Name |
Tacrolimus |
Valganciclovir |
|
Mechanism 2
|
Nephrotoxicity |
Nephrotoxicity |
| Key Mechanism Factor 2 |
| Factor Name |
Nephrotoxicity |
| Factor Description |
The combination of drugs that can induce nephrotoxicity may increase the risk of kidney injury. When kidney injury occurs, the inability to remove excess urine and waste from the body can lead to high levels of urea nitrogen, creatinine, and electrolytes (such as potassium and magnesium) in the blood. |
| Mechanism Description |
- Increased risk of nephrotoxicity by the combination of Tacrolimus and Valganciclovir
|


