Details of Drug-Drug Interaction
| Drug General Information (ID: DDIZ4LNYXP) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Clevidipine | Drug Info | Nitrous acid | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antihypertensives Agent | Antidotes | |||||||
| Structure | |||||||||
| Mechanism of Clevidipine-Nitrous acid Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
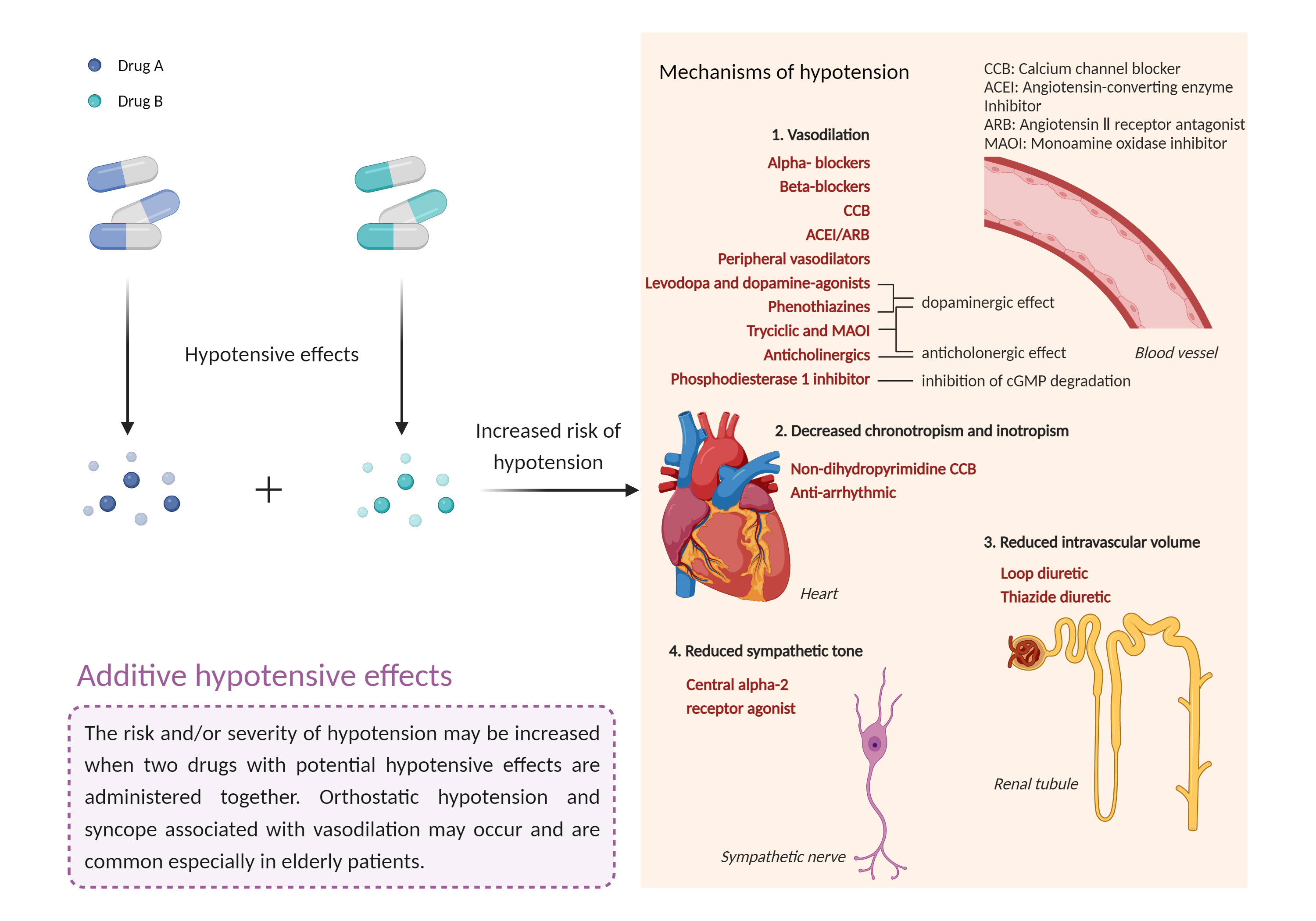
| Additive hypotensive effects Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Clevidipine | Nitrous acid | |||||||
| Mechanism |
Hypotensive effects Calcium channel unspecific Blocker |
Hypotensive effects Guanylate cyclase Agonist |
|||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Voltage-dependent L-type calcium channel | Structure Sequence | |||||||
| Protein Family | Calcium channel beta subunit family | ||||||||
| Protein Function |
Regulatory subunit of L-type calcium channels (PubMed:1309651, PubMed:8107964, PubMed:15615847). Regulates the activity of L-type calcium channels that contain CACNA1A as pore-forming subunit (By similarity). Regulates the activity of L-type calcium channels that contain CACNA1C as pore-forming subunit and increases the presence of the channel complex at the cell membrane (PubMed:15615847). Required for functional expression L-type calcium channels that contain CACNA1D as pore-forming subunit (PubMed:1309651). Regulates the activity of L-type calcium channels that contain CACNA1B as pore-forming subunit (PubMed:8107964).
Click to Show/Hide
|
||||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Guanylate cyclase soluble | Structure Sequence | |||||||
| Protein Family | Adenylyl cyclase class-4/guanylyl cyclase family | ||||||||
| Protein Function |
There are two types of guanylate cyclases: soluble forms and membrane-associated receptor forms. Activated by nitric oxide in the presence of magnesium or manganese ions.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Sodium nitrite should be used with caution in the presence of concomitant antihypertensive agents, diuretics, vasodilators, or PDE5 inhibitors. Hemodynamics should be monitored during and after administration of sodium nitrite, and the infusion rate decreased if significant hypotension occurs. In addition, methemoglobin levels should be monitored and oxygen administered during treatment whenever possible. | ||||||||
