Details of Drug-Drug Interaction
| Drug General Information (ID: DDIXQW8B2H) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Isotretinoin | Drug Info | Remdesivir | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antiacne Agents/Antineoplastics | Purine Nucleosides | |||||||
| Structure | |||||||||
| Mechanism of Isotretinoin-Remdesivir Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
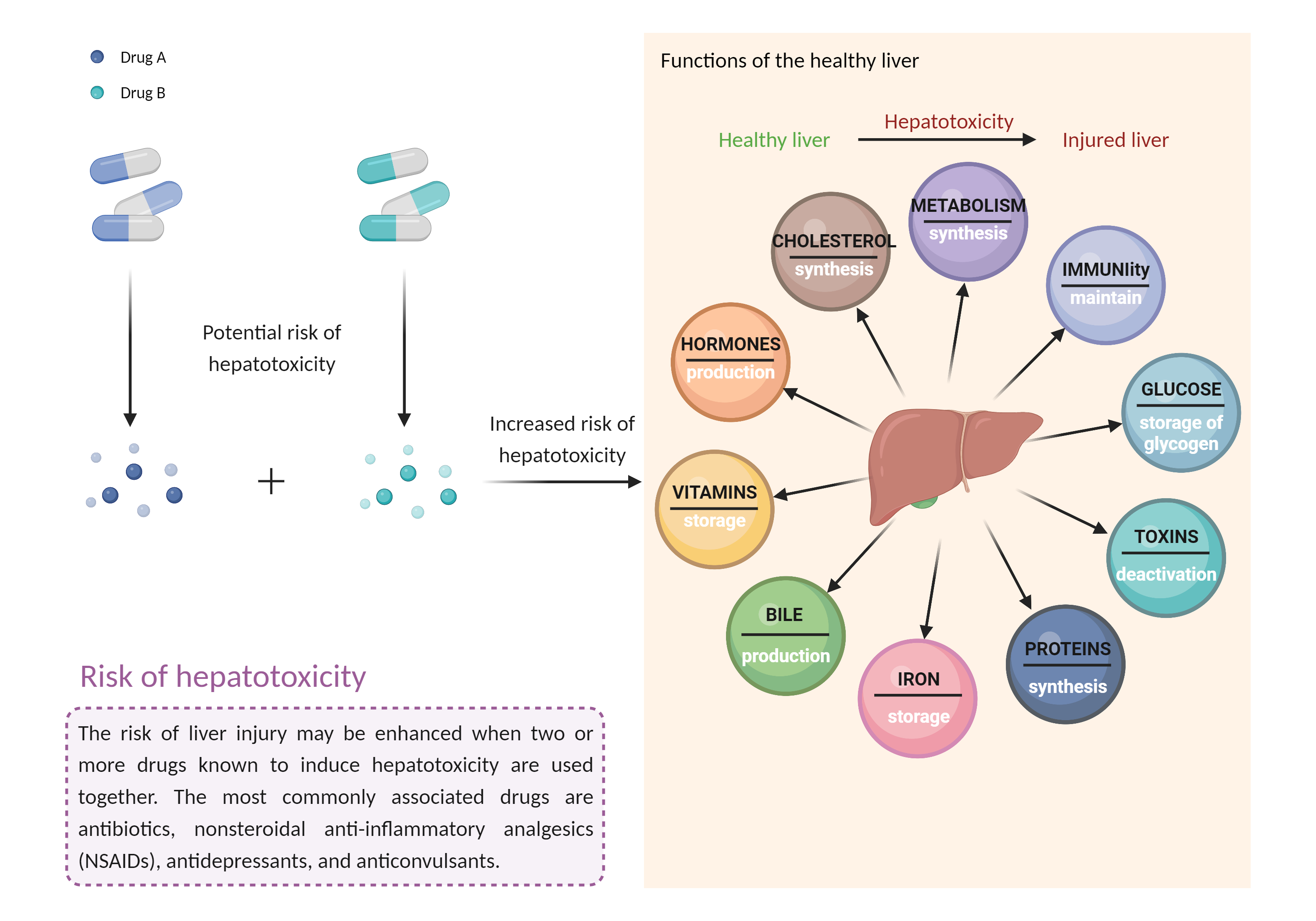
| Increased risk of hepatotoxicity Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Isotretinoin | Remdesivir | |||||||
| Mechanism | Hepatotoxicity | Hepatotoxicity | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Hepatotoxicity | ||||||||
| Factor Description | Combination of drugs that can induce hepatotoxicity may increase the risk of liver injury. Symptoms vary depending on the level of exposure and the total extent of liver damage, and may cause few symptoms if the damage is mild, and eventually lead to liver failure in patients with severe damage. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Until more information is available, concomitant use of remdesivir with known hepatotoxic drugs should be avoided when possible. Liver function should be evaluated prior to starting remdesivir and monitored during treatment as clinically appropriate. Remdesivir should not be initiated in patients with ALT greater than or equal to 5 times the upper limit of normal (ULN) at baseline. Likewise, remdesivir should be discontinued immediately if ALT increases to 5 times ULN or greater during treatment (may be restarted when ALT falls below 5 times ULN), or if ALT elevation is accompanied by signs or symptoms of liver inflammation or increasing conjugated bilirubin, alkaline phosphatase, or INR. | ||||||||
