Details of Drug-Drug Interaction
| Drug General Information (ID: DDIW6H2ADB) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Quinidine | Drug Info | Sodium acetate | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Group I Antiarrhythmics | Minerals And Electrolytes | |||||||
| Structure | |||||||||
| Mechanism of Quinidine-Sodium acetate Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
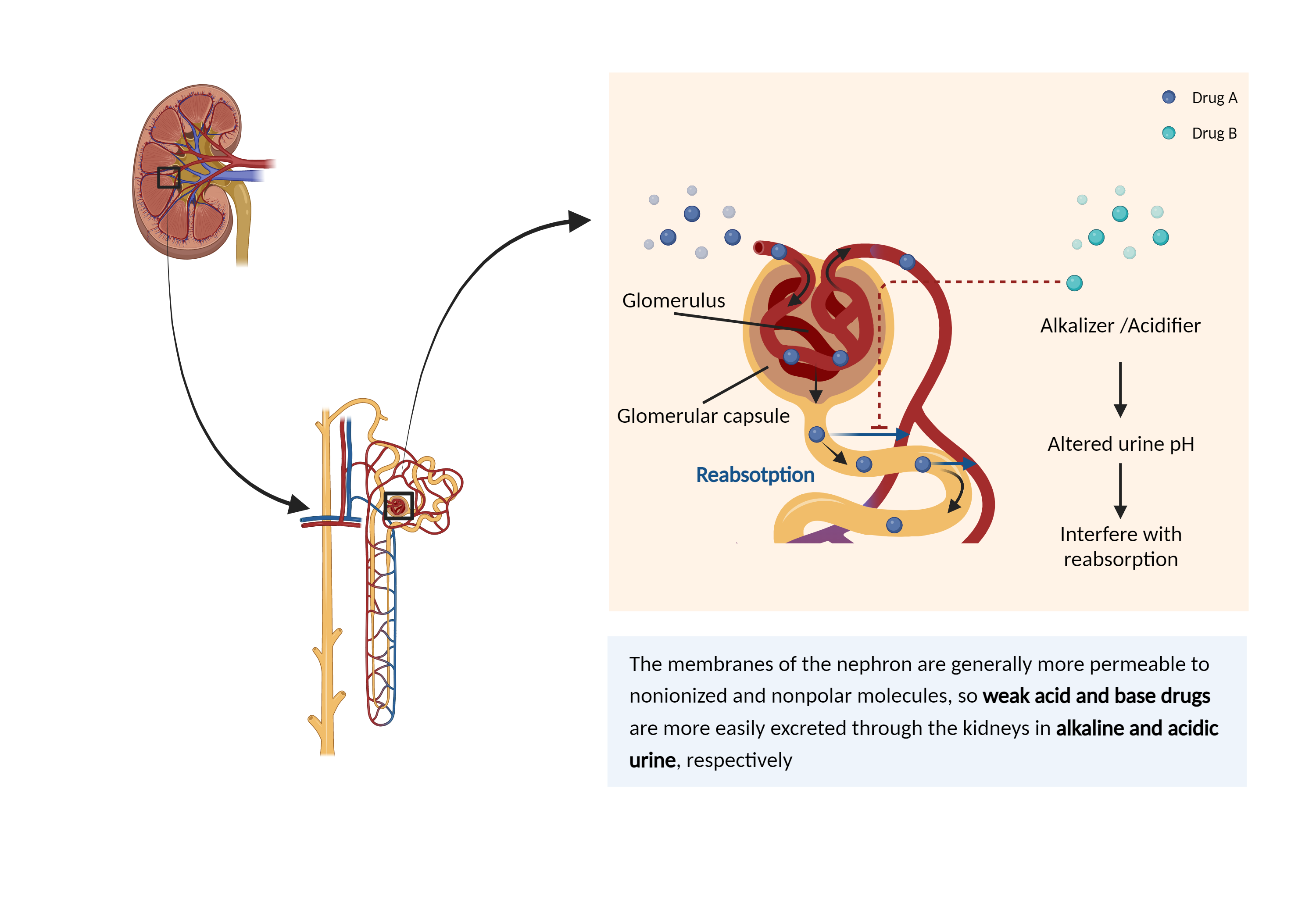
| Altered urine pH Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Quinidine | Sodium acetate | |||||||
| Mechanism | Urine pH sensitive | Urine alkalinizer | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Urine pH | ||||||||
| Factor Description | The normal range of urine pH is between 4.5 and 8, depending mainly on the hydrogen ion concentration. Changes in urine pH may significantly affect drug reabsorption and excretion. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised if quinidine is used with agents that can increase urinary pH. Quinidine levels may need to be monitored more closely following addition or discontinuation of these agents, and the quinidine dosage adjusted as necessary. Patients should be advised to seek medical attention if they experience symptoms that could indicate quinidine toxicity such as tinnitus, hearing loss, visual disturbances, diarrhea, headache, dizziness, palpitations, syncope, or irregular heartbeats. | ||||||||
