Details of Drug-Drug Interaction
| Drug General Information (ID: DDIVGUQ819) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Talimogene laherparepvec | Drug Info | Brexucabtagene autoleucel | Drug Info | |||||
| Drug Type | Vaccine | Autologous cell transplant | |||||||
| Therapeutic Class | Anticancer Agents | Antineoplastics | |||||||
| Mechanism of Talimogene laherparepvec-Brexucabtagene autoleucel Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
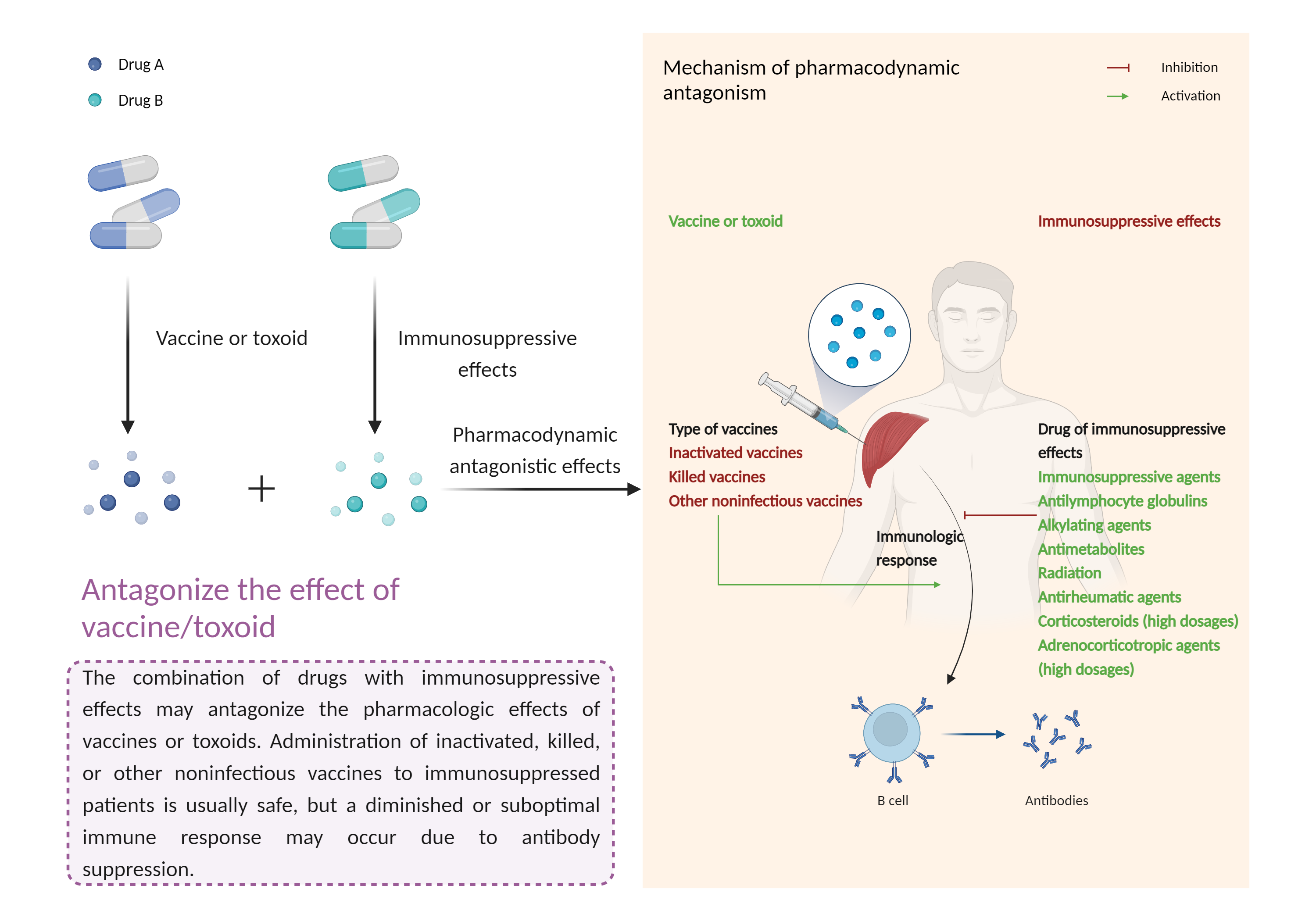
| Antagonize the effect of vaccine/toxoid Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Talimogene laherparepvec | Brexucabtagene autoleucel | |||||||
| Mechanism | Immunomodulatory infective agent | Immunosuppressive effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Vaccine/toxoid | ||||||||
| Factor Description | The beneficial effects of the vaccine/toxinoid may be reduced, leading to a diminished or suboptimal immune response. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Talimogene laherparepvec should not be used in patients receiving immunosuppressive therapy or cancer chemotherapy. Treatment with talimogene laherparepvec may need to be deferred until after such therapy is discontinued and the patient's immune system has sufficiently recovered. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Imlygic (talimogene laherparepvec). Amgen USA, Thousand Oaks, CA. | ||||||||||||||||||
