| Mechanism of Tramadol-Pimavanserin Interaction
(Severity Level: Major)
|
|
Additive CNS depression effects
Click to Show/Hide Mechanism Graph
|
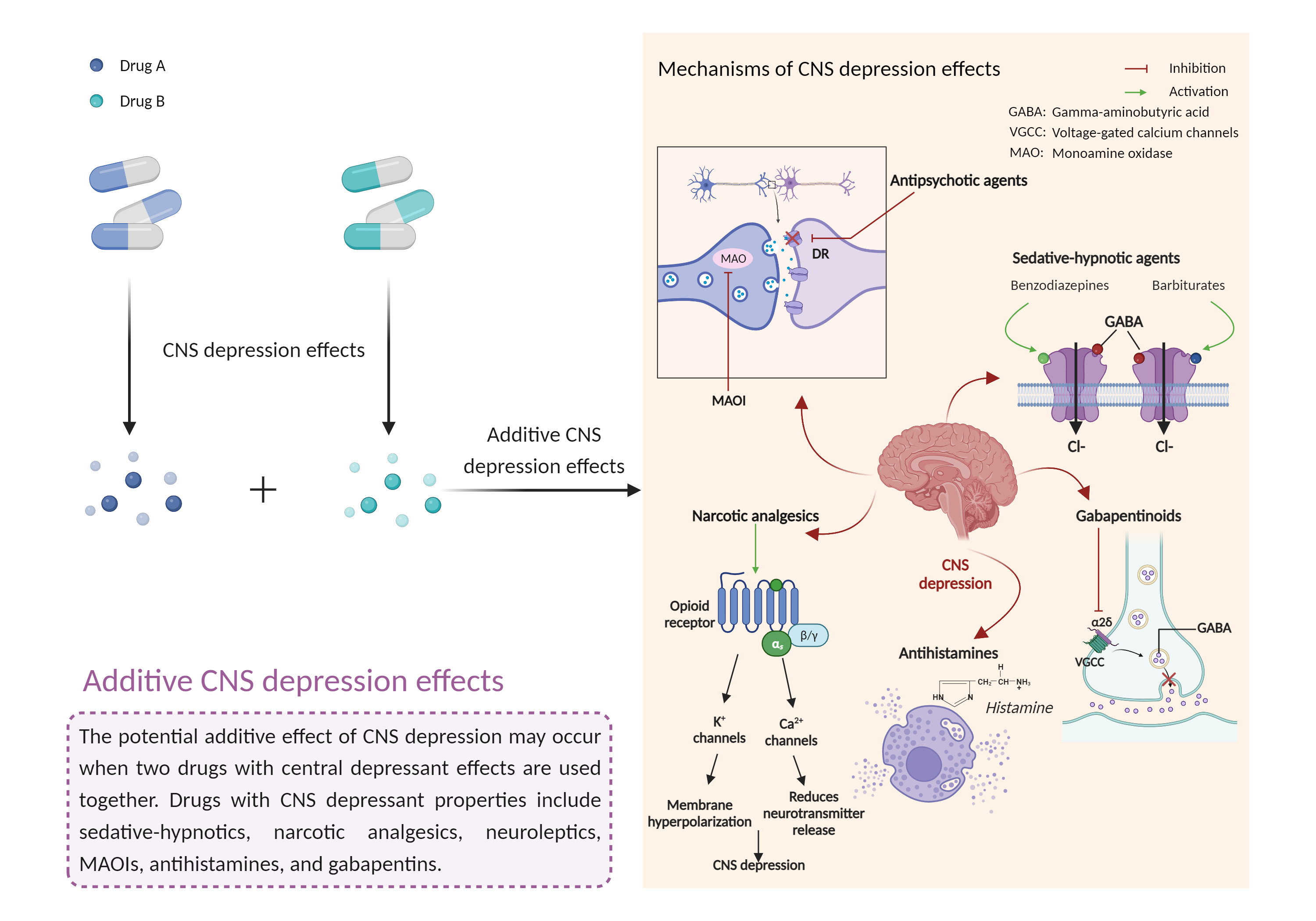 |
| Drug Name |
Tramadol |
Pimavanserin |
|
Mechanism 1
|
CNS depression effects |
CNS depression effects |
| Key Mechanism Factor 1 |
| Factor Name |
CNS depression effects |
| Factor Description |
CNS depressants are drugs that inhibit or suppress brain activity and can reduce mental and physical processes. Excessive CNS depression can lead to decreased heart rate, slow breathing (less than 10 breaths per minute), extreme confusion or loss of memory, nausea and vomiting, poor judgment, blue lips or fingertips, irritability and aggression, and clammy or cold skin. |
| Mechanism Description |
- Additive CNS depression effects by the combination of Tramadol and Pimavanserin
|
|
Increased risk of prolong QT interval
Click to Show/Hide Mechanism Graph
|
 |
| Drug Name |
Tramadol |
Pimavanserin |
|
Mechanism 2
|
Prolong QT interval |
Prolong QT interval |
| Key Mechanism Factor 2 |
| Factor Name |
QT interval |
| Factor Description |
Long QT syndrome is a heart signaling disorder that can cause a fast, chaotic heartbeat (arrhythmia). Many people may not exhibit symptoms, and usually the condition is detected during routine medical tests. In others, the most common symptoms include: sudden fainting, palpitations, dizziness, seizures, sudden death. |
| Mechanism Description |
- Increased risk of prolong QT interval by the combination of Tramadol and Pimavanserin
|


