Details of Drug-Drug Interaction
| Drug General Information (ID: DDISQY8OM7) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Esomeprazole | Drug Info | Tofacitinib | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antiulcer Agents | Antirheumatics | |||||||
| Structure | |||||||||
| Mechanism of Esomeprazole-Tofacitinib Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| CYP450 enzyme inhibition Click to Show/Hide Mechanism Graph | |||||||||
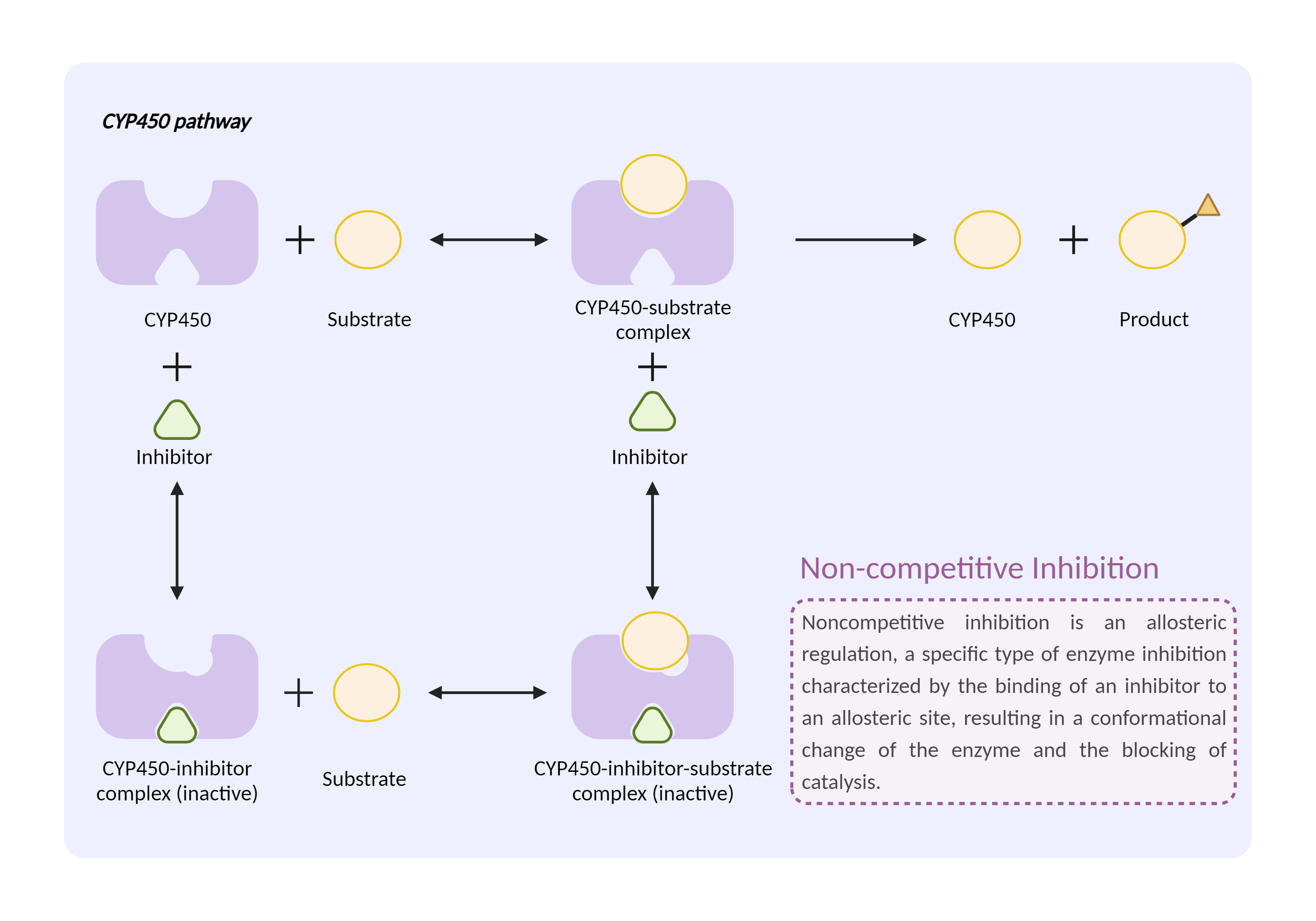 |
|||||||||
| Drug Name | Esomeprazole | Tofacitinib | |||||||
| Mechanism | CYP450 2C19 inhibitor | CYP450 2C19 substrate | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Cytochrome P450 2C19 |
×
Structure
Sequence
MDPFVVLVLCLSCLLLLSIWRQSSGRGKLPPGPTPLPVIGNILQIDIKDVSKSLTNLSKIYGPVFTLYFGLERMVVLHGYEVVKEALIDLGEEFSGRGHFPLAERANRGFGIVFSNGKRWKEIRRFSLMTLRNFGMGKRSIEDRVQEEARCLVEELRKTKASPCDPTFILGCAPCNVICSIIFQKRFDYKDQQFLNLMEKLNENIRIVSTPWIQICNNFPTIIDYFPGTHNKLLKNLAFMESDILEKVKEHQESMDINNPRDFIDCFLIKMEKEKQNQQSEFTIENLVITAADLLGAGTETTSTTLRYALLLLLKHPEVTAKVQEEIERVIGRNRSPCMQDRGHMPYTDAVVHEVQRYIDLIPTSLPHAVTCDVKFRNYLIPKGTTILTSLTSVLHDNKEFPNPEMFDPRHFLDEGGNFKKSNYFMPFSAGKRICVGEGLARMELFLFLTFILQNFNLKSLIDPKDLDTTPVVNGFASVPPFYQLCFIPV
|
|||||||
| Gene Name | CYP2C19 | ||||||||
| Uniprot ID | CP2CJ_HUMAN | ||||||||
| KEGG Pathway | hsa:1557 | ||||||||
| Protein Family | Cytochrome P450 family | ||||||||
| Protein Function |
A cytochrome P450 monooxygenase involved in the metabolism of polyunsaturated fatty acids (PUFA) (PubMed:18577768, PubMed:19965576, PubMed:20972997). Mechanistically, uses molecular oxygen inserting one oxygen atom into a substrate, and reducing the second into a water molecule, with two electrons provided by NADPH via cytochrome P450 reductase (NADPH--hemoprotein reductase) (PubMed:18577768, PubMed:19965576, PubMed:20972997). Catalyzes the hydroxylation of carbon-hydrogen bonds. Hydroxylates PUFA specifically at the omega-1 position (PubMed:18577768). Catalyzes the epoxidation of double bonds of PUFA (PubMed:20972997, PubMed:19965576). Also metabolizes plant monoterpenes such as limonene. Oxygenates (R)- and (S)-limonene to produce carveol and perillyl alcohol (PubMed:11950794). Responsible for the metabolism of a number of therapeutic agents such as the anticonvulsant drug S-mephenytoin, omeprazole, proguanil, certain barbiturates, diazepam, propranolol, citalopram and imipramine. Hydroxylates fenbendazole at the 4' position (PubMed:23959307).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | The dosage of tofacitinib should be reduced by 50% when used concomitantly with one or more medications that may result in both moderate inhibition of CYP450 3A4 and potent inhibition of CYP450 2C19. For example, the dose for patients receiving 10 mg twice daily should be reduced to 5 mg twice daily and the dose for patients receiving 5 mg twice daily should be reduced to 5 mg once daily. For patients receiving 11 mg once daily of the extended-release formulation, the dose should be reduced to 5 mg once daily of the immediate-release formulation. The dose for patients receiving 3.2 mg twice daily should be reduced to 3.2 mg once daily and the dose for patients receiving 4 mg twice daily should be reduced to 4 mg once daily. Moderate inhibitors of CYP450 3A4 include amiodarone, aprepitant, ciprofloxacin, crizotinib, darunavir, dalfopristin-quinupristin, diltiazem, dronedarone, erythromycin, fluconazole, fusidic acid, grapefruit juice, imatinib, isavuconazonium, netupitant, and verapamil. Potent inhibitors of CYP450 2C19 include fluconazole, fluvoxamine, esomeprazole, lansoprazole, and omeprazole. Inhibitors of CYP450 2C19 alone are unlikely to substantially alter the pharmacokinetics of tofacitinib. | ||||||||
