Details of Drug-Drug Interaction
| Drug General Information (ID: DDIS4X7U1E) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Clozapine | Drug Info | Inotuzumab ozogamicin | Drug Info | |||||
| Drug Type | Small molecule | Monoclonal antibody | |||||||
| Therapeutic Class | Antipsychotic Agents | Antineoplastics | |||||||
| Mechanism of Clozapine-Inotuzumab ozogamicin Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
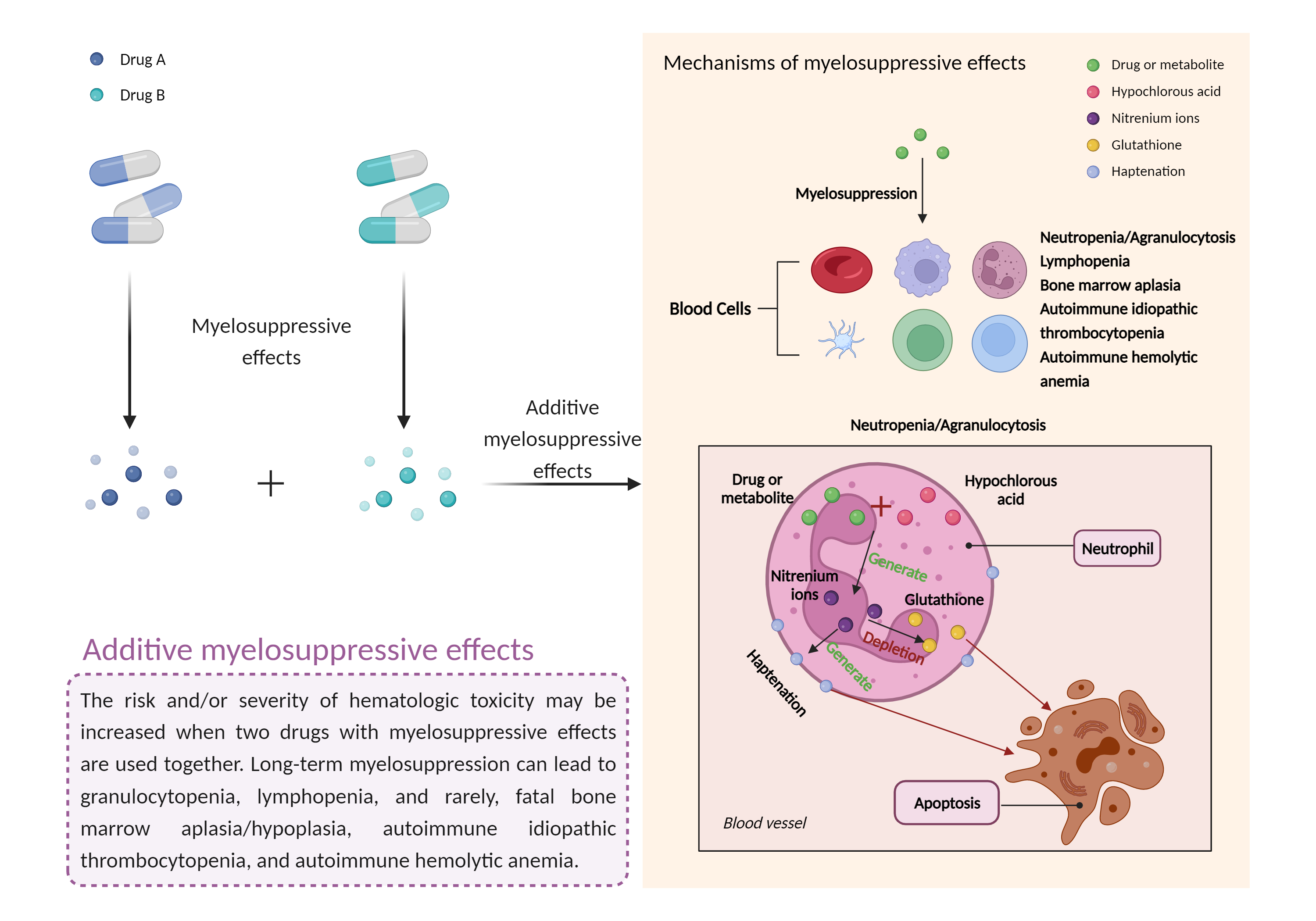
| Additive myelosuppressive effects Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Clozapine | Inotuzumab ozogamicin | |||||||
| Mechanism 1 | Hematologic toxicity | Hematologic toxicity | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Myelosuppressive effects | ||||||||
| Factor Description | Myelosuppression, also known as bone marrow suppression, is a decrease in bone marrow activity that leads to a decrease in the production of blood cells. Some blood cell disorders include: erythrocytopenia (anemia), leukopenia (neutropenia), and thrombocytopenia (thrombocytopenia). | ||||||||
| Mechanism Description |
|
||||||||
| Increased risk of prolong QT interval Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Clozapine | Inotuzumab ozogamicin | |||||||
| Mechanism 2 | Prolong QT interval | Prolong QT interval | |||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | QT interval | ||||||||
| Factor Description | Long QT syndrome is a heart signaling disorder that can cause a fast, chaotic heartbeat (arrhythmia). Many people may not exhibit symptoms, and usually the condition is detected during routine medical tests. In others, the most common symptoms include: sudden fainting, palpitations, dizziness, seizures, sudden death. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution and close monitoring are advised when clozapine is used with other agents that have a well-known potential to cause agranulocytosis or otherwise suppress bone marrow function, such as antineoplastic, antimalarial, and antirheumatic agents. Caution is recommended if clozapine is used in combination with other drugs that can prolong the QT interval. Serum electrolytes, including potassium, magnesium and calcium, should be measured at baseline and periodically during treatment, and any abnormalities corrected prior to initiating clozapine. Clozapine treatment should be discontinued if the QTc interval exceeds 500 msec. Patients should be advised to seek prompt medical attention if they experience symptoms that could indicate the occurrence of torsade de pointes such as dizziness, lightheadedness, fainting, palpitation, irregular heart rhythm, shortness of breath, or syncope. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Clozaril (clozapine). Novartis Pharmaceuticals, East Hanover, NJ. | ||||||||||||||||||
| 2 | Product Information. Tasigna (nilotinib). Novartis Pharmaceuticals, East Hanover, NJ. | ||||||||||||||||||
