Details of Drug-Drug Interaction
| Drug General Information (ID: DDIS4X68UF) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Carteolol (ophthalmic) | Drug Info | Flecainide | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antihypertensive Agents | Antiarrhythmic Agents | |||||||
| Structure | |||||||||
| Mechanism of Carteolol (ophthalmic)-Flecainide Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
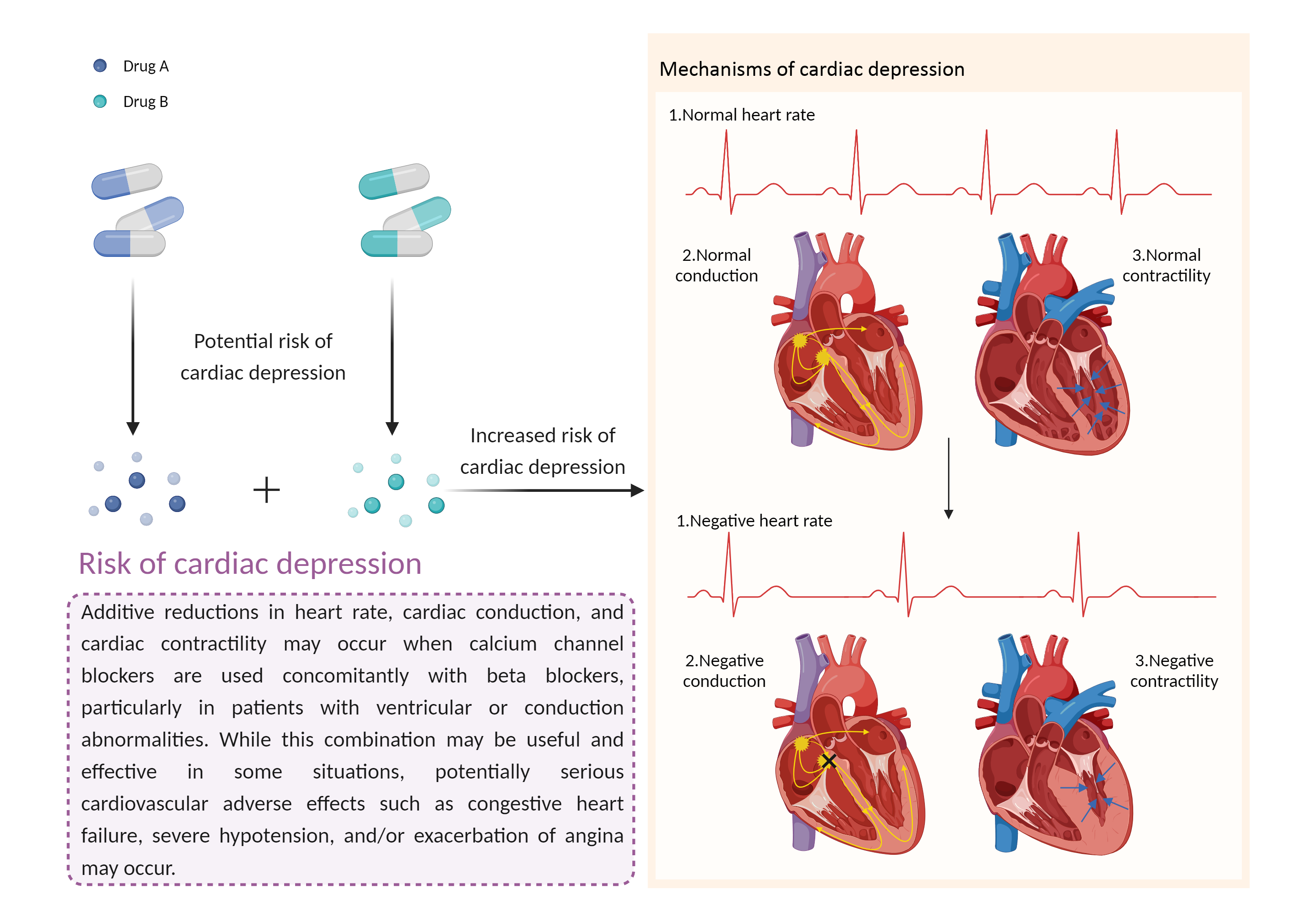
| Increased risk of cardiac depression Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Carteolol (ophthalmic) | Flecainide | |||||||
| Mechanism | Cardiac depressant effects | Cardiac depressant effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Cardiac depression | ||||||||
| Factor Description | Cardiac depression may manifest as negative heart rate, negative conduction, and negative contractility. Symptoms include: excessive or more than normal sleep, irritability, loss of interest in activities, and fatigue. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Careful monitoring of the patient's hemodynamic status is recommended during concomitant administration. The same precaution should be observed when beta blocker ophthalmic solutions are used, since they are systemically absorbed and can produce clinically significant systemic effects even at low or undetectable plasma levels. Patients should be advised to promptly report symptoms such as dizziness, slow or irregular heartbeats, syncope, or palpitations. | ||||||||
